Abstract
Summary
This is a case of a patient with a 10-year history of Cushing’s disease (CD) that was previously treated with transsphenoidal pituitary tumour resection. Conception occurred spontaneously, and during early pregnancy recurrent CD became apparent both clinically and biochemically. Repeat transsphenoidal surgery took place during the second trimester, and the high-risk pregnancy resulted in a live neonate. Despite evidence of hypercortisolism and recurrent CD at 6 months postpartum, the patient had a second successful, uncomplicated pregnancy, further adding to the rarity and complexity of this case. Pregnancy in CD is rare because hypercortisolism seen in CD suppresses gonadotropin release, leading to menstrual irregularities and infertility. Diagnosis of CD is particularly challenging because many clinical and biochemical features of normal pregnancy overlap considerably with those seen in CD. Diagnosis and treatment are extremely important to reduce rates of perinatal morbidity and mortality.
Learning points
- Hypercortisolism suppresses gonadotropin release, leading to menstrual irregularities and infertility. In CD, hypersecretion of both androgens and cortisol further contributes to higher rates of amenorrhoea and infertility.
- Pregnancy itself is a state of hypercortisolism, with very few studies detailing normal ranges of cortisol in each trimester of pregnancy for midnight salivary cortisol and urinary free cortisol testing.
- Treatment of CD reduces maternal morbidity and rates of foetal loss, and can be either surgical (preferred) or medical.
- CD can relapse, often many years after initial surgery.
- There are a limited number of cases of Cushing’s syndrome in pregnancy, therefore, the best possible treatment is difficult to determine and should be individualised to the patient.
Background
CD is rare in the general population. It is even rarer to present as a clinical conundrum during pregnancy. Diagnosis is challenging due to the overlap of physiological hormonal changes during pregnancy with features of Cushing’s syndrome, and it is further complicated by limited data for cortisol reference ranges in a pregnant state. The prognostic benefits of treatment of CD in pregnancy in reducing perinatal morbidity and mortality must be carefully weighed up against the risks of surgery and/or medical management in pregnancy.
Case presentation
The patient was a 31-year-old female diagnosed with Cushing’s disease at age 20 years. Initial clinical features were oligomenorrhoea, weight gain, hypertension, and impaired glucose tolerance. She had markedly elevated 24 h urinary free cortisol (UFC) of 1,984 nmol/day, which was six times the upper limit of normal (ULN). Results of a 1 mg dexamethasone suppression test (DST) showed failure to suppress cortisol levels, with an elevated morning cortisol of 695 nmol/L (reference range (RR): 100–690). ACTH levels remained inappropriately normal at 7.3 pmol/L (RR: < 12.1), suggesting ACTH-dependent hypercortisolism. A 5 mm by 4.4 mm microadenoma was identified on magnetic resonance imaging (MRI) scan of the pituitary gland, and she underwent initial transsphenoidal pituitary adenectomy. Histopathological examination demonstrated positive staining for adrenocorticotrophic hormone (ACTH). Immediately after surgery, she required hydrocortisone and levothyroxine replacement for several months, which was gradually weaned and eventually ceased. She had routine MRI with gadolinium and biochemical surveillance for 5 years, which showed cortisol levels within the normal ranges and no visible pituitary lesion on imaging, and she was then lost to follow-up. Results of 1 mg DST and 24 h UFC measurements were not available from this time period. Other medical history was significant for mild depression. The patient was a non-smoker and did not drink alcohol.
At age 30 years, the patient experienced weight gain and facial rounding, prompting an endocrinology referral. While awaiting review, she spontaneously achieved conception and was confirmed to be 6 weeks’ gestation at time of the first visit. An early diagnosis of gestational diabetes mellitus was made, and she commenced insulin therapy. Gestational hypertension was also confirmed, treated with methyldopa 500 mg mane and 250 mg midi. Other medications included folic acid 5 mg daily, cholecalciferol, and ferrous sulphate.
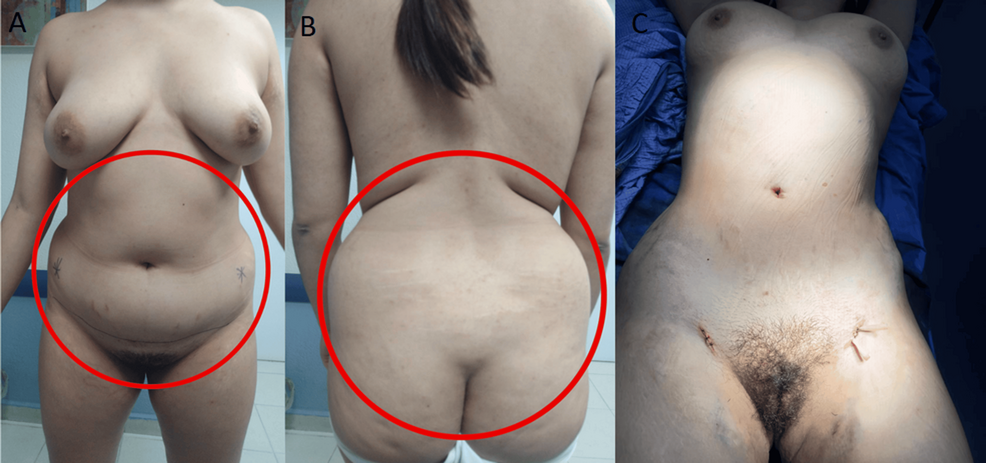
The patient was referred to a tertiary hospital high-risk pregnancy service for ongoing care. She was initially reviewed at 8 + 5 weeks’ gestation and was noted to have plethora, round facies, and prominent dorsocervical fat pads. Central adiposity with violaceous striae over the lower abdomen was evident. Visual fields were normal to gross confrontation, and formal visual field assessment was confirmed to be normal. Weight was 70 kg, with BMI 26.7 kg/m2.
As pregnancy progressed, insulin and antihypertensive requirements increased, with an additional methyldopa 250 mg nocte required to keep blood pressure at target.
Investigation
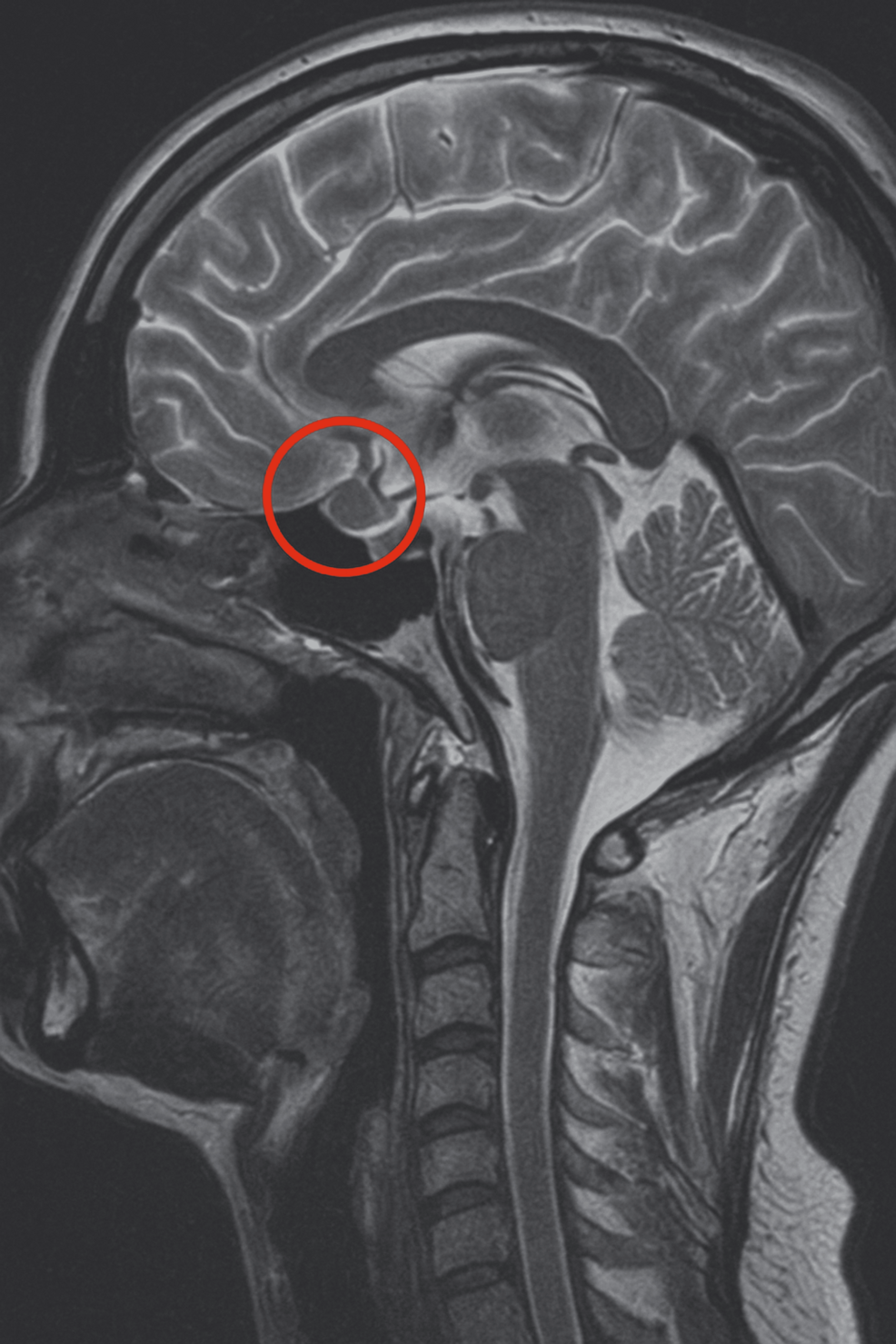
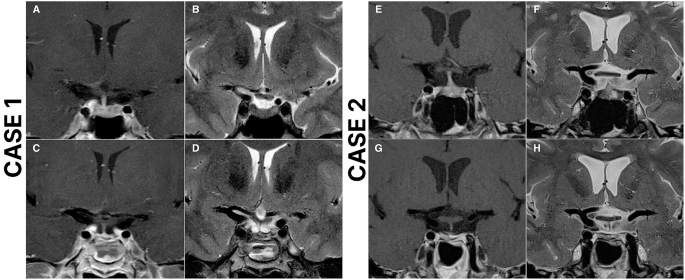
The 24 h UFC was 450 nmol/24 h (1.5× ULN of non-pregnant reference range). Late-night salivary cortisol (LNSC) was 17 nmol/L (non-pregnant reference range <8 nmol/L). Serum pathology results are shown in Table 1. MRI brain performed at 6 weeks’ gestation revealed a possible 6 by 4 mm nodule in the left lateral aspect of the sella (Fig. 1). IV contrast was not used as the patient was within the first trimester.
| Investigation | Result | Reference range |
|---|---|---|
| Fasting glucose, mmol/L | 5.2 | |
| HbA1c, % | 5.4 | |
| 24 h urinary cortisol, nmol/d | 450 | 54–319 |
| Cortisol (08:22), nmol/L | 521 | 138–650 |
| Midnight salivary cortisol, nmol/L | 17 | <8 |
| ACTH, pmol/L | 10 | <12.1 |
| IGF-1, nmol/L | 31 | 12–42 |
| Growth hormone, mIU/L | 2.9 | 0–15 |
| TSH, mIU/L | 2.34 | 0.4–3.2 |
| FT4, pmol/L | 11.9 | 11–17 |

Citation: Endocrinology, Diabetes & Metabolism Case Reports 2025, 4; 10.1530/EDM-25-0092
At 14 weeks’ gestation, the repeat 24 h UFC was 542 nmol/L and LNSC was 17 nmol. There is a lack of pregnancy-specific reference ranges for 24 h UFC or LNSC measurements, making it difficult to make a definitive biochemical diagnosis. After careful discussion in a multidisciplinary team meeting, she proceeded with bilateral inferior petrosal sinus sampling (IPSS), which demonstrated a central to peripheral gradient with values presented in Table 2.
| 0 | 2 min | 5 min | 10 min | 15 min | |
|---|---|---|---|---|---|
| Right | 258 | 823 | 1,040 | 864 | 728 |
| Left | 73 | 196 | 228 | 263 | 234 |
| Peripheral | 12 | 41 | 56 | 81 | 86 |
| Right: peripheral | 21.50 | 20.07 | 18.57 | 10.67 | 8.46 |
| Left: peripheral | 6.08 | 4.78 | 4.07 | 3.25 | 2.72 |
Treatment
The patient underwent transsphenoidal resection of her adenoma at 17+ weeks’ gestation. She recovered uneventfully.
Day 1 postoperative cortisol level remained elevated at 706 nmol/L, falling to 587 nmol/L by Day 3. Postoperative steroid treatment was not required.
Histopathological examination demonstrated a pituitary adenoma with mild nuclear atypia and infrequent positive ACTH staining (Fig. 2). In addition to the tumour and normal pituitary tissue, there was also abundant eosinophilic proteinaceous material present, which may have suggested contents of an associated cyst, although presence of cyst lining was not present to confirm this diagnosis. A small fragment of included bone appeared invaded by the adenoma within the resected tissue.

Citation: Endocrinology, Diabetes & Metabolism Case Reports 2025, 4; 10.1530/EDM-25-0092
Outcome and follow-up
The patient’s insulin and antihypertensive requirements plateaued postoperatively. Serial ultrasound showed that the fetal size was consistently in the 15th percentile. There were no features of preeclampsia throughout gestation.
At 35 + 5 weeks’ gestation, she had premature rupture of membranes and delivered a healthy live male infant weighing 2,250 g via normal vaginal delivery. Diabetes and hypertension resolved promptly after delivery, with cessation of insulin and antihypertensive medications.
At 5 weeks postpartum morning cortisol was within normal range at 265 nmol/L, with ACTH 6.8 pmol/L. At 10 weeks postpartum, the 24 h UFC was within normal limits at 136 nmol/day, and a 1 mg DST showed a detectable, equivocal cortisol level of 98 nmol/L. Repeat MRI pituitary was performed 2 months postpartum, which did not show any residual pituitary adenoma. No pituitary hormone replacement was required.
By 6 months postpartum, repeat 1 mg DST showed failure to suppress cortisol, with cortisol level at 154 nmol/L (RR without dexamethasone: 138–650 nmol/L), suggesting residual CD. Ambulatory blood pressure monitoring revealed essential hypertension, with average BP 141/101 mmHg across 24 h, requiring treatment with methyldopa. Despite evidence of residual CD, the patient desired a second pregnancy. Reassuringly, her cortisol burden was low, with LNSC 5 nmol/L (RR: < 8) and 24 h UFC 143 nmol/day (non-pregnant RR: 54–319), both within reference range. No definite lesion was identified on MRI brain with intravenous contrast. Extensive discussions between the endocrinologist, maternal–foetal medicine specialist, neurosurgeon, and the patient were held. The pros and cons of pursuing further treatment such as radiotherapy versus proceeding with pregnancy despite suggestion of active Cushing’s disease were explicitly discussed.
The patient confirmed her second pregnancy 11 months after the birth of her first child, and this proceeded without complications. There was no evidence of gestational diabetes on 75 g glucose tolerance tests performed at 16 and 26 weeks’ gestation. Blood pressure was well managed on methyldopa alone. She delivered a healthy male infant via normal vaginal delivery at 38 weeks’ gestation and breastfed successfully. MRI was performed at 16 weeks postpartum and did not show an appreciable sella/suprasellar mass. Repeat 24 h UFC was 275 nmol/day, consistent with ongoing CD. Clinical features of CD returned, included central adiposity, liver function test derangement, and raised HbA1c with fasting hyperinsulinaemia. Pituitary radiation therapy was discussed, including the possibility of more than one dose being required, the strong likelihood of inducing panhypopituitarism, and the unknown duration of time between radiation and remission (1). The alternative option of medical management with osilodrostat was discussed, given its recent availability and government subsidy in Australia. The patient was recently commenced on osilodrostat 1 mg twice daily after ECG attendance to exclude prolonged QTc, and patient education regarding the potential risk of hypoadrenalism and when to seek medical attention.
Discussion
Managing Cushing’s disease (CD) in pregnancy is complex and requires a multidisciplinary approach, as recurrence can occur years after initial remission. Suspected Cushing’s syndrome (CS) requires careful assessment. In cases where active disease poses significant maternal and foetal risks, transsphenoidal pituitary surgery can be safely performed in the second trimester. CD increases the risk of gestational diabetes and hypertension, requiring close monitoring to optimise outcomes. Postpartum, persistent hypercortisolism may indicate recurrence, highlighting the need for long-term endocrine follow-up. Despite mild residual disease, successful pregnancies are possible with appropriate monitoring and management, emphasising the importance of thorough family planning discussions.
UFC values are twice as high in pregnant patients compared to non-pregnant controls (2). In the first trimester of normal pregnancy, UFC values are normal, but by the third trimester, they increase three-fold up to values seen in CS (3). Therefore, CS should only be suspected when UFC values in the second and third trimesters are greater than three times the upper limit of normal (3). LNSC is a useful screening test because in CS, the usual circadian nadir of cortisol secretion is lost. At least 2–3 UFC and/or NSC screening tests are recommended (4). Lopes et al. (5) established reference values for LNSC with suggested normal ranges of 0.8–6.9 nmol/L in the first trimester, 1.1–7.2 nmol/L in the second trimester, and 1.9–9.1 nmol/L in the third trimester (5). The use of a 1 mg DST in pregnancy is not recommended because the hypothalamus–pituitary–adrenal (HPA) axis response to exogenous glucocorticoids is blunted, making it difficult to interpret test results (3).
Adrenal adenomas are responsible for 40–50% of CS cases in pregnancy, while CD causes 33% (3). In non-pregnant patients, ACTH levels are useful to classify the likely cause of CS. Undetectable ACTH levels cannot be relied upon for diagnosis in pregnancy because ACTH levels are elevated in pregnancy (3). Using high-dose dexamethasone suppression testing (HDST) as an initial test in pregnant patients has been recommended (3). A lack of suppression of ACTH with administration of high-dose dexamethasone suggests adrenal aetiology. However, HDST is not always definitive (3). Ultrasound imaging of the adrenal glands is also recommended as an initial investigation because most adrenal lesions can be visualised (3, 5). Pregnancy complicates visualisation of a pituitary mass by MRI imaging because physiologic enlargement of the pituitary gland during pregnancy may mask small tumours (6). Non-contrast MRI has reduced sensitivity for detection of CD. However, gadolinium contrast is not recommended in pregnant women (7).
Inferior petrosal sinus sampling (IPSS) is the gold standard for diagnosing CD in the non-pregnant population (4). The most recent guidelines for diagnosis of CS suggest that IPSS is not necessary for diagnosis if MRI clearly shows a tumour >10 mm in the context of dynamic test results and clinical features that also strongly suggest CD (4). Lindsay and colleagues (3) caution the use of IPSS unless prior non-invasive testing remains equivocal due to risks of thromboembolism and exposure to radiation posed by IPSS (3). However, these risks can be mitigated with extra precautions during pregnancy, including use of lead barrier protection, a direct jugular approach, and with the procedure occurring at a specialised centre (3).
Treatment of CS in pregnancy should be individualised depending on the patient presentation and gestational age (4). Active treatment of CS, by either medical or surgical intervention, reduces maternal morbidity and rates of foetal loss (4). Surgery is usually preferred because there are fewer complications at delivery and it has high rates of remission (8). Surgery reduces rates of perinatal and maternal morbidity but does not reduce rates of preterm birth and intrauterine growth restriction (IUGR) (9). Pituitary or adrenal surgery should ideally be done in the second trimester, before week 24 of pregnancy, in a high-volume centre with multidisciplinary team input (8). There is a risk of spontaneous abortion with anaesthesia given in the first trimester and an increased risk of premature labour with anaesthesia given in the third trimester (7).
Unfortunately, CD can recur, and 50% of recurrence occurs within 50 months of pituitary surgery (1, 4). Recurrence is defined as ongoing clinical and biochemical evidence of hypercortisolism after an initial period of remission. Factors that increase the likelihood of postoperative remission included the identification of a tumour on MRI pre-surgery, no invasion of the sinus cavernous by the adenoma, older age (greater than 35 years), low postoperative cortisol and ACTH levels, and long-term hypocortisolism (greater than 1 year) (1). A second pituitary surgery is often the first-line treatment option in recurrence, which has overall lower rates of remission compared to first surgery and increased risk of hypopituitarism due to scar tissue in the pituitary and often the need for more aggressive surgical technique (1). Both fractionated radiotherapy and stereotactic radiosurgery are therapeutic options and achieve high rates of remission (1).
There are no medications that are approved for treatment of CD in pregnancy, although the latest guidelines suggest consideration of metyrapone, ketoconazole, or cabergoline (4, 6). The newer agent, osilodrostat inhibits the enzymes 11-beta-hydroxylase and 18-hydroxylase, reducing production of cortisol and aldosterone respectively, thereby normalising UFC values, reducing systolic and diastolic blood pressure, fasting blood glucose levels, and improving body weight in clinical trials (10). There is no information on osilodrostat use and safety in pregnancy, but it is an effective agent in patients who are unsuitable for surgery and patients with recurrent disease after surgery (10). It is associated with risk of hypoadrenalism, prolongation of the QTc interval, and increased serum testosterone levels, particularly at higher doses (10). Each medication poses its own risk of side effects and therefore treatment must be individualised. Overall, medical treatment should only be used in pregnancy when surgical treatment is contraindicated (6).
Our case demonstrates a rare case of CD in pregnancy with no significant adverse perinatal outcomes for mother or baby, albeit late preterm delivery in the first pregnancy. Ongoing endocrinology surveillance is essential to monitor for recurrent CD.
Declaration of interest
The authors declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Funding
This research did not receive any specific grant from any funding agency in the public, commercial, or not-for-profit sector.
Patient consent
Written informed consent for publication of their clinical details was obtained from the patient.
Author contribution statement
Several case details and timeline of events were gathered by EW. This is a patient of SG.
References
-
1↑Braun L , Rubinstein G , Zopp S , et al. Recurrence after pituitary surgery in adult Cushing’s disease: a systematic review on diagnosis and treatment. Endocrine 2020 70 218–231. (https://doi.org/10.1007/s12020-020-02432-z)
-
2↑Odagiri E , Ishiwatari N , Abe Y , et al. Hypercortisolism and the resistance to dexamethasone suppression during gestation. Endocrinol Jpn 1988 35 685–690. (https://doi.org/10.1507/endocrj1954.35.685)
-
3↑Lindsay J , Jonklaas J , Oldfield E , et al. Cushing’s syndrome during pregnancy: personal experience and review of the literature. J Clin Endocrinol Metab 2005 90 3077–3083. (https://doi.org/10.1210/jc.2004-2361)
-
4↑Fleseriu M , Auchus R , Bancos I , et al. Consensus on diagnosis and management of Cushing’s disease: a guideline update. Lancet Diabetes Endocrinol 2021 9 847–875. (https://doi.org/10.1016/s2213-8587(21)00235-7)
-
5↑Lopes L , Francisco R , Galletta M , et al. Determination of nighttime salivary cortisol during pregnancy: comparison with values in non-pregnancy and Cushing’s disease. Pituitary 2016 19 30–38. (https://doi.org/10.1007/s11102-015-0680-3)
-
6↑Lindsay J & Nieman L . The hypothalamic-pituitary-adrenal axis in pregnancy: challenges in disease detection and treatment. Endocr Rev 2005 26 775–799. (https://doi.org/10.1210/er.2004-0025)
-
7↑Abbassy M , Kshettry V , Hamrahian A , et al. Surgical management of recurrent Cushing’s disease in pregnancy: a case report. Surg Neurol Int 2015 6 S640–S645. (https://doi.org/10.4103/2152-7806.170472)
-
8↑Brue T , Amodru V & Castinetti F . Management of Cushing’s syndrome during pregnancy: solved and unsolved questions. Eur J Endocrinol 2018 178 259–266. (https://doi.org/10.1530/eje-17-1058)
-
9↑Caimari F , Valassi E , Garbayo P , et al. Cushing’s syndrome and pregnancy outcomes: a systematic review of published cases. Endocrine 2017 55 555–563. (https://doi.org/10.1007/s12020-016-1117-0)
-
10↑Perosevic M & Tritos NA . Clinical utility of osilodrostat in Cushing’s disease: review of currently available literature. Drug Des Devel Ther 2023 17 1303–1312. (https://doi.org/10.2147/dddt.s315359)
Filed under: Cushing's, pituitary, Rare Diseases, Treatments | Tagged: pituitary, pregnancy, recurrence, transsphenoidal | Leave a comment »