Highlights
- •
-
Cushing syndrome (CS) is a rare disorder with a variety of underlying etiologies.
- •
-
CS is expected to affect 0.2 to 5 people per million per year.
- •
-
Adrenal-dependent CS is an uncommon variant of CS.
- •
-
This study reports a rare occurrence of pituitary and adrenal adenoma with CS.
Abstract
Introduction
Cushing syndrome is a rare disorder with a variety of underlying etiologies, that can be exogenous or endogenous (adrenocorticotropic hormone (ACTH)-dependent or ACTH-independent). The current study aims to report a case of ACTH-independent Cushing syndrome with adrenal adenoma and nonfunctioning pituitary adenoma.
Case report
A 37–year–old female presented with amenorrhea for the last year, associated with weight gain. She had a moon face, buffalo hump, and central obesity. A 24-hour urine collection for cortisol was performed, revealing elevated cortisol. Cortisol level was non-suppressed after administering dexamethasone. MRI of the pituitary revealed a pituitary microadenoma, and the CT scan of the abdomen with adrenal protocol revealed a left adrenal adenoma.
Discussion
Early diagnosis may be postponed due to the variety of clinical presentations and the referral of patients to different subspecialists based on their dominant symptoms (gynecological, dermatological, cardiovascular, psychiatric); it is, therefore, critical to consider the entire clinical presentation for correct diagnosis.
Conclusion
Due to the diversity in the presentation of CS, an accurate clinical, physical and endocrine examination is always recommended.
Keywords
1. Introduction
Cushing syndrome (CS) is a collection of clinical manifestations caused by an excess of glucocorticoids [1]. CS is a rare disorder with a variety of underlying etiologies that can be exogenous due to continuous corticosteroid therapy for any underlying inflammatory illness or endogenous due to either adrenocorticotropic hormone (ACTH)-dependent or ACTH-independent [2], [3]. Cushing syndrome is expected to affect 0.2 to 5 people per million per year. Around 10% of such cases involve children [4], [5]. ACTH-dependent glucocorticoid excess owing to pituitary adenoma accounts for the majority (60–70%) of endogenous CS, with primary adrenal causes accounting for only 20–30% and ectopic ACTH-secreting tumors accounting for the remaining 5–10% [6]. Adrenal-dependent CS is an uncommon variant of CS caused mostly by benign (90%) or malignant (8%) adrenal tumors or, less frequently, bilateral micronodular (1%) or macronodular (1%) adrenal hyperplasia [7].
The current study aims to report a case of ACTH-independent Cushing syndrome with adrenal adenoma and nonfunctioning pituitary adenoma. The report has been arranged in line with SCARE guidelines and includes a brief literature review [8].
2. Case report
2.1. Patient’s information
A 37–year–old female presented with amenorrhea for the last year, associated with weight gain. She denied having polyuria, polydipsia, headaches, visual changes, dizziness, dryness of the skin, cold intolerance, or constipation. She had no history of chronic disease and denied using steroids. She visited an internist, a general surgeon, and a gynecologist and was treated for hypothyroidism. She was put on Thyroxin 100 μg daily, and oral contraceptive pills were given for her menstrual problems. Last time, the patient was referred to an endocrinology clinic, and they reviewed the clinical and physical examinations.
2.2. Clinical examination
She had a moon face, buffalo hump, central obesity, pink striae over her abdomen, and proximal weakness of the upper limbs. After reviewing the history and clinical examination, CS was suspected.
2.3. Diagnostic assessment
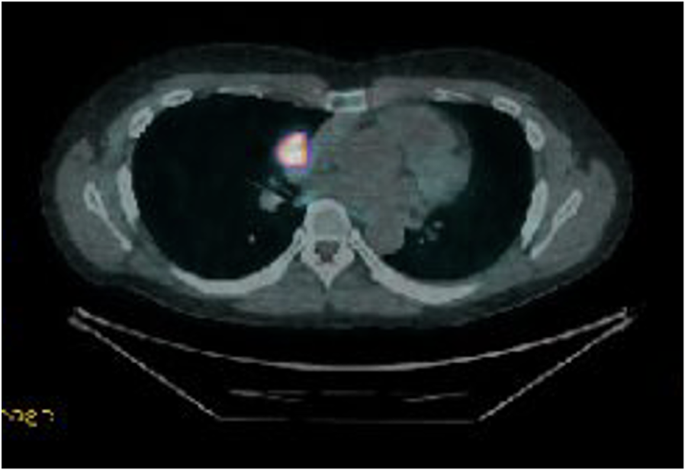
Because the thyroid function test revealed low thyroid-stimulating hormone (TSH), free T3, and freeT4, the patient was sent for a magnetic resonance imaging (MRI) of the pituitary, which revealed a pituitary microadenoma (7 ∗ 6 ∗ 5) mm (Fig. 1). Since the patient was taking thyroxin and oral contraceptive pills, the investigations were postponed for another six weeks due to the contraceptive pills’ influence on the results of the hormonal assessment for CS. After six weeks of no medication, a 24-hour urinary free cortisol (UFC) was performed three times, revealing elevated cortisol levels (1238, 1100, and 1248) nmol (normal range, 100–400) nmol. A dexamethasone suppression test was done (after administering dexamethasone tab 1 mg at 11 p.m., serum cortisol was measured at 9 a.m.). The morning serum cortisol level was 620 nmol (non-suppressed), which normally should be less than 50 nmol. The ACTH level was below 1 pg/mL.

Fig. 1. Contrast enhanced T1W weighted MRI (coronal section) showing small 7 mm hypo-enhanced microadenoma (yellow arrow) in right side of pituitary gland with mild superior bulge.
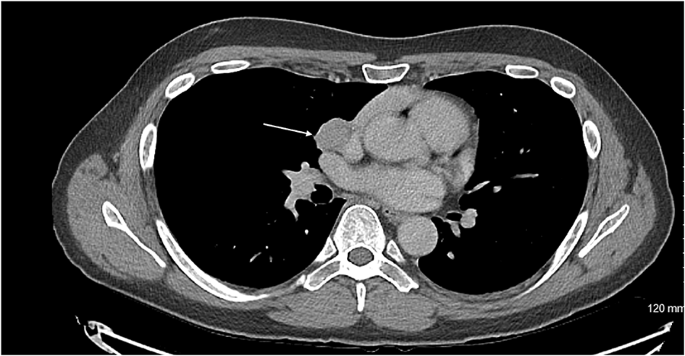
Based on these findings, ACTH independent CS was suspected. The computerized tomography (CT) scan of the abdomen with adrenal protocol revealed a left adrenal adenoma (33 mm × 25 mm) without features of malignancy (Fig. 2).

Fig. 2. Computed tomography scan of the abdomen with IV contrast, coronal section, showing 33 mm × 25 mm lobulated enhanced left adrenal tumor (yellow arrow), showing absolute washout on dynamic adrenal CT protocol, consistent with adrenal adenoma.
2.4. Therapeutic intervention
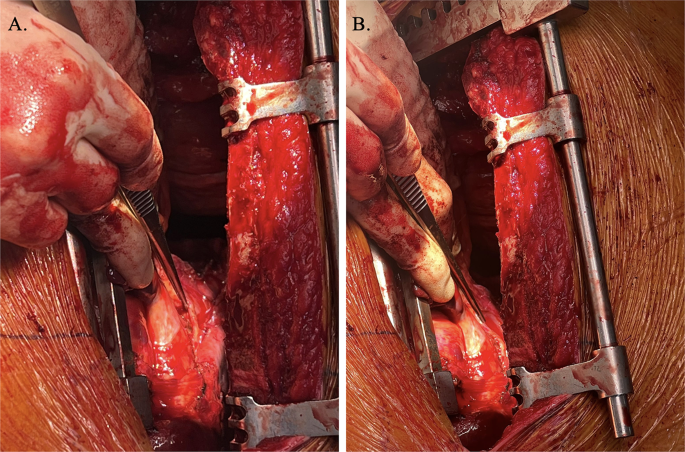
The patient was referred to the urologist clinic for left adrenalectomy after preparation for surgery and perioperative hormonal management. She underwent laparoscopic adrenalectomy and remained in the hospital for two days. The histopathology results supported the diagnosis of adrenal adenoma.
2.5. Follow-up
She was released home after two days on oral hydrocortisone 20 mg in the morning and 10 mg in the afternoon. After one month of follow-up, serum cortisol was 36 nmol, with the resolution of some features such as weight reduction (3 kg) and skin color (pink striae became white).
3. Discussion
Cushing’s syndrome is a serious and well-known medical condition that results from persistent exposure of the body to excessive glucocorticoids, either from endogenous or, most frequently, exogenous sources [9]. The average age of diagnosis is 41.4 years, with a female-to-male ratio of 3:1 [10]. ACTH-dependent CS accounts for almost 80% of endogenous CS, while ACTH-independent CS accounts for nearly 20% [10]. This potentially fatal condition is accompanied by several comorbidities, including hypertension, diabetes, coagulopathy, cardiovascular disease, infections, and fractures [11]. Exogenous CS, also known as iatrogenic CS, is more prevalent than endogenous CS and is caused by the injection of supraphysiologic glucocorticoid dosages [12]. ACTH-independent CS is induced by uncontrolled cortisol release from an adrenal gland lesion, most often an adenoma, adrenocortical cancer, or, in rare cases, ACTH-independent macronodular adrenal hyperplasia or primary pigmented nodular adrenal disease [13].
The majority of data suggests that early diagnosis is critical for reducing morbidity and mortality. Detection is based on clinical suspicion initially, followed by biochemical confirmation [14]. The clinical manifestation of CS varies depending on the severity and duration of glucocorticoid excess [14]. Some individuals may manifest varying symptoms and signs because of a rhythmic change in cortisol secretion, resulting in cyclical CS [15]. The classical symptoms of CS include weight gain, hirsutism, striae, plethora, hypertension, ecchymosis, lethargy, monthly irregularities, diminished libido, and proximal myopathy [16]. Neurobehavioral presentations include anxiety, sadness, mood swings, and memory loss [17]. Less commonly presented features include headaches, acne, edema, abdominal pain, backache, recurrent infection, female baldness, dorsal fat pad, frank diabetes, electrocardiographic abnormalities suggestive of cardiac hypertrophy, osteoporotic fractures, and cardiovascular disease from accelerated atherosclerosis [10]. The current case presented with amenorrhea, weight gain, moon face, buffalo hump, and skin discoloration of the abdomen.
Similar to the current case, early diagnosis may be postponed due to the variety of clinical presentations and the referral of patients to different subspecialists based on their dominant symptoms (gynecological, dermatological, cardiovascular, psychiatric); it is, therefore, critical to consider the entire clinical presentation for correct diagnosis [18]. Weight gain may be less apparent in children, but there is frequently an arrest in growth with a fall in height percentile and a delay in puberty [19].
The diagnosis and confirmation of the etiology can be difficult and time-consuming, requiring a variety of laboratory testing and imaging studies [20]. According to endocrine society guidelines, the initial assessment of CS must include one or more of the three following tests: 24-hour UFC measurement; evaluation of the diurnal variation of cortisol secretion by assessing the midnight serum or salivary cortisol level; and a low-dose dexamethasone suppression test, typically the 1 mg overnight test [21]. Although UFC has sufficient sensitivity and specificity, it does not function well in milder cases of Cushing’s syndrome [22]. In CS patients, the typical circadian rhythm of cortisol secretion is disrupted, and a high late-night cortisol serum level is the earliest and most sensitive diagnostic indicator of the condition [23]. In the current case, the UFC was elevated, and cortisol was unsuppressed after administration of dexamethasone.
All patients with CS should have a high-resolution pituitary MRI with a gadolinium-based contrast agent to prove the existence or absence of a pituitary lesion and to identify the source of ACTH between pituitary adenomas and ectopic lesions [24]. Adrenal CT scan is the imaging modality of choice for preoperatively localizing and subtyping adrenocortical lesions in ACTH-independent Cushing’s syndrome [9]. MRI of the pituitary gland of the current case showed a microadenoma and a CT scan of the adrenals showed left adrenal adenoma.
Surgical resection of the origin of the ACTH or glucocorticoid excess (pituitary adenoma, nonpituitary tumor-secreting ACTH, or adrenal tumor) is still the first-line treatment of all forms of CS because it leaves normal adjacent structures and results in prompt remission and inevitable recovery of regular adrenal function [12], [25]. Laparoscopic (retroperitoneal or transperitoneal) adrenalectomy has become the gold standard technique for adrenal adenomas since it is associated with fewer postoperative morbidity, hospitalization, and expense when compared to open adrenalectomy [17]. In refractory cases, or when a patient is not a good candidate for surgery, cortisol-lowering medication may be employed [26]. The current case underwent left adrenalectomy.
Symptoms of CS, such as central obesity, muscular wasting or weakness, acne, hirsutism, and purple striae generally improve first and may subside gradually over a few months or even a year; nevertheless, these symptoms may remain in 10–30% of patients [27]. Glucocorticoid replacement is essential after adrenal-sparing curative surgery until the pituitary-adrenal function returns, which might take up to two years, especially if adrenal adenomas have been resected [25]. Chronic glucocorticoid excess causes lots of new co-morbidities, lowering the quality of life and increasing mortality. The most common causes of mortality in CS are cardiovascular disease and infections [28]. After one month of follow-up, serum cortisol was 36 nmol, and several features, such as weight loss (3 kg) and skin color, were resolved (pink striae became white).
In conclusion, the coexistence of adrenal adenoma and pituitary adenoma with CS is a rare possibility. Due to the diversity in the presentation of CS, an accurate clinical, physical and endocrine examination is always recommended. Laparoscopic adrenalectomy is the gold standard for treating adrenal adenoma.
Consent
Written informed consent was obtained from the patient’s family for publication of this case report and accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal on request.
Provenance and peer review
Not commissioned, externally peer-reviewed.
Ethical approval
Approval is not necessary for case report (till 3 cases in single report) in our locality.
The family gave consent for the publication of the report.
Funding
None.
Guarantor
Fahmi Hussein Kakamad, Fahmi.hussein@univsul.edu.iq.
Research registration number
Not applicable.
CRediT authorship contribution statement
-
Abdulwahid M. Salh: major contribution of the idea, literature review, final approval of the manuscript.
-
Rawa Bapir: Surgeon performing the operation, final approval of the manuscript.
-
Fahmi H. Kakamad: Writing the manuscript, literature review, final approval of the manuscript.
-
Soran H. Tahir, Fattah H. Fattah, Aras Gh. Mahmood, Rawezh Q. Salih, Shaho F. Ahmed: literature review, final approval of the manuscript.
Declaration of competing interest
None to be declared.
References
- [1]
-
S.M. Ahmed, S.F. Ahmed, S. Othman, B.A. Abdulla, S.H. Mohammed, A.M. Salih, et al.Topical corticosteroid-induced iatrogenic Cushing syndrome in an infant; a case report with literature reviewAnn.Med.Surg., 71 (2021), Article 102978
- [2]
-
L.H. Broersen, C.D. Andela, O.M. Dekkers, A.M. Pereira, N.R. BiermaszImprovement but no normalization of quality of life and cognitive functioning after treatment of Cushing syndromeJ.Clin.Endocrinol.Metab., 104 (11) (2019), pp. 5325-5337
- [3]
-
R.L. Hopkins, M.C. LeinungExogenous Cushing’s syndrome and glucocorticoid withdrawalEndocrinol. Metab. Clin., 34 (2) (2005), pp. 371-384
- [4]
-
D. Hirsch, G. Tsvetov, Y. Manisterski, N. Aviran-Barak, V. Nadler, S. Alboim, et al.Incidence of Cushing’s syndrome in patients with significant hypercortisoluriaEur. J. Endocrinol., 176 (1) (2017), pp. 41-48
- [5]
-
B. Bista, N. BeckCushing syndromeIndian J.Pediatr., 81 (2) (2014 Feb), pp. 158-164
- [6]
-
M. Lodish, C.A. StratakisA genetic and molecular update on adrenocortical causes of Cushing syndromeNat. Rev. Endocrinol., 12 (5) (2016), pp. 255-262
- [7]
-
J.M. Swain, C.S. Grant, R.T. Schlinkert, G.B. Thompson, R.V. Lloyd, W.F. YoungCorticotropin-independent macronodular adrenal hyperplasia: a clinicopathologic correlationArch. Surg., 133 (5) (1998), pp. 541-546
- [8]
-
R.A. Agha, T. Franchi, C. Sohrabi, G. Mathew, for the SCARE GroupThe SCARE 2020 guideline: updating consensus Surgical CAse REport (SCARE) guidelinesInt. J. Surg., 84 (2020), pp. 226-230
- [9]
-
K. Duan, K.G. Hernandez, O. MeteRepublished: clinicopathological correlates of adrenal Cushing’s syndromePostgrad. Med. J., 91 (1076) (2015 Jun 1), pp. 331-342
- [10]
-
A. Lacroix, R.A. Feelders, C.A. Stratakis, L.K. NiemanCushing’s syndromeLancet, 386 (9996) (2015), pp. 913-927
- [11]
-
L.K. Nieman, B.M. Biller, J.W. Findling, J. Newell-Price, M.O. Savage, P.M. Stewart, et al.The diagnosis of Cushing’s syndrome: an endocrine society clinical practice guidelineJ.Clin.Endocrinol.Metab., 93 (5) (2008), pp. 1526-1540
- [12]
-
S.T. Sharma, L.K. Nieman, R.A. FeeldersCushing’s syndrome: epidemiology and developments in disease managementClin.Epidemiol., 7 (2015), p. 281
- [13]
-
J. Newell-Price, X. Bertagna, A.B. Grossman, L.K. NiemanCushing’s syndromeLancet, 367 (9522) (2006), pp. 1605-1617
- [14]
-
L.K. NiemanCushing’s syndrome: update on signs, symptoms and biochemical screeningEur. J. Endocrinol., 173 (4) (2015), pp. M33-M38
- [15]
-
B. Atkinson, K.R. MullanWhat is the best approach to suspected cyclical Cushing syndrome? Strategies for managing Cushing’s syndrome with variable laboratory dataClin. Endocrinol., 75 (1) (2011), pp. 27-30
- [16]
-
F. Ferrau, M. KorbonitsMetabolic comorbidities in Cushing’s syndromeEur. J. Endocrinol., 173 (4) (2015), pp. M133-M157
- [17]
-
V. NeychevCushing syndrome: presentation, diagnosis, and treatment, including subclinical Cushing syndromeManagement of Adrenal Masses in Children And Adults, Springer, Cham (2017), pp. 159-178
- [18]
-
L.K. NiemanDiagnosis of Cushing’s syndrome in the modern eraEndocrinol. Metab. Clin., 47 (2) (2018), pp. 259-273
- [19]
-
H.L. Storr, L.F. Chan, A.B. Grossman, M.O. SavagePaediatric Cushing’s syndrome: epidemiology, investigation and therapeutic advancesTrendsEndocrinol.Metab., 18 (4) (2007), pp. 167-174
- [20]
-
R. Pivonello, A.M. Isidori, M.C. De Martino, J. Newell-Price, B.M. Biller, A. ColaoComplications of Cushing’s syndrome: state of the artLancet DiabetesEndocrinol., 4 (7) (2016), pp. 611-629
- [21]
-
C. Tatsi, C.A. StratakisCushing disease: diagnosis and treatmentPituitary Disorders of Childhood, Humana Press, Cham (2019), pp. 89-114
- [22]
-
K.I. Alexandraki, A.B. GrossmanIs urinary free cortisol of value in the diagnosis of Cushing’s syndrome?Curr.Opin.Endocrinol.DiabetesObes., 18 (4) (2011), pp. 259-263
- [23]
-
E.M. Cardoso, A.L. Arregger, O.R. Tumilasci, L.N. ContrerasDiagnostic value of salivary cortisol in Cushing’s syndrome (CS)Clin. Endocrinol., 70 (4) (2009), pp. 516-521
- [24]
-
G. Arnaldi, A. Angeli, A.B. Atkinson, X. Bertagna, F. Cavagnini, G.P. Chrousos, et al.Diagnosis and complications of Cushing’s syndrome: a consensus statementJ.Clin.Endocrinol.Metab., 88 (12) (2003), pp. 5593-5602
- [25]
-
L.K. Nieman, I. IliasEvaluation and treatment of Cushing’s syndromeAm. J. Med., 118 (12) (2005), pp. 1340-1346
- [26]
-
N.A. Wagner-Bartak, A. Baiomy, M.A. Habra, S.V. Mukhi, A.C. Morani, B.R. Korivi, et al.Cushing syndrome: diagnostic workup and imaging features, with clinical and pathologic correlationAm. J. Roentgenol., 209 (1) (2017), pp. 19-32
- [27]
-
R.A. Feelders, S.J. Pulgar, A. Kempel, A.M. PereiraManagement of endocrine disease: the burden of Cushing’s disease: clinical and health-related quality of life aspectsEur. J. Endocrinol., 167 (3) (2012), pp. 311-326
- [28]
-
A. Ferriere, A. TabarinCushing’s syndrome: treatment and new therapeutic approachesBest Pract. Res. Clin. Endocrinol. Metab., 34 (2) (2020), Article 101381
Filed under: adrenal, Cushing, Cushing's | Tagged: ACTH, adrenal, pituitary | Leave a comment »