Abstract
Summary
ACTH-secreting pituitary adenomas causing Cushing’s disease (CD) typically present with weight gain, whereas weight loss and hypokalemia in endogenous Cushing’s patients are suggestive of ectopic ACTH production. We report a case of CD presenting with atypical features of marked weight loss and hypokalemia. A 75-year-old female was admitted to the hospital with a history of profound weight loss, associated with uncontrolled hypertension, hyperglycemia, severe proximal muscle weakness, and hypokalemia. Subsequent investigations, including 24-h urinary free cortisol, 48-h low-dose dexamethasone suppression test, MRI of the sella, and bilateral inferior petrosal sinus sampling, confirmed CD without any evidence of ectopic ACTH production. She became eucortisolemic with medical therapy of ketoconazole and cabergoline, subsequently regained her weight, and became normokalemic. This case illustrates that patients with CD may present with symptoms and biochemical findings that would otherwise suggest ectopic ACTH production.
Learning points
- Patients with CD do not always present with classical clinical features and may present with symptoms and biochemical findings that would otherwise suggest ectopic ACTH production.
- While most patients with CD typically lose weight after biochemical remission, some patients gain weight after the normalization of cortisol levels.
- This case highlights the need to entertain a broad differential in patients presenting with hypokalemia and weight loss and the need to exclude hypercortisolemia.
Background
Pituitary corticotropin (ACTH)-induced Cushing’s disease (CD) accounts for approximately 70% of patients presenting with Cushing’s syndrome (1). ACTH-producing pituitary adenomas are typically microadenomas and, in over a third of CD patients, there is no demonstrable lesion on MRI (2). Clinical and biochemical diagnosis of CD may be challenging, as patients can present with varied symptoms that overlap with other comorbidities. Progressive weight gain associated with central adiposity is a common manifestation of CD occurring during the early stage of the disease. While nonspecific features such as hypertension, diabetes, cardiac hypertrophy, arterial and venous thrombosis, electrolyte abnormalities, and psychiatric disturbances also occur frequently, the more discriminatory signs of hypercortisolemia include proximal myopathy, facial plethora, easy bruising, and wide striae (2). Weight loss with associated hypokalemia typically suggests an underlying ectopic ACTH production. Here we report an unusual case of pituitary ACTH-induced CD who presented with significant hypokalemia and marked weight loss which resolved with medical control of CD.
Case presentation
A 75-year-old female with a history of type 2 diabetes, hypertension, osteoporosis, and coronary artery disease presented to the emergency department (ED) with profound proximal muscle weakness associated with a serum potassium of 2.4 mmol/L (normal = 3.6–5.2 mmol/L). She also reported a weight loss of 90 lbs over the previous 2 years. In addition, she had uncontrolled hypertension despite taking three anti-hypertensive agents and worsening glycemic control requiring increasing anti-hyperglycemic therapy; her hemoglobin A1c at presentation was 9.3%.
Investigation
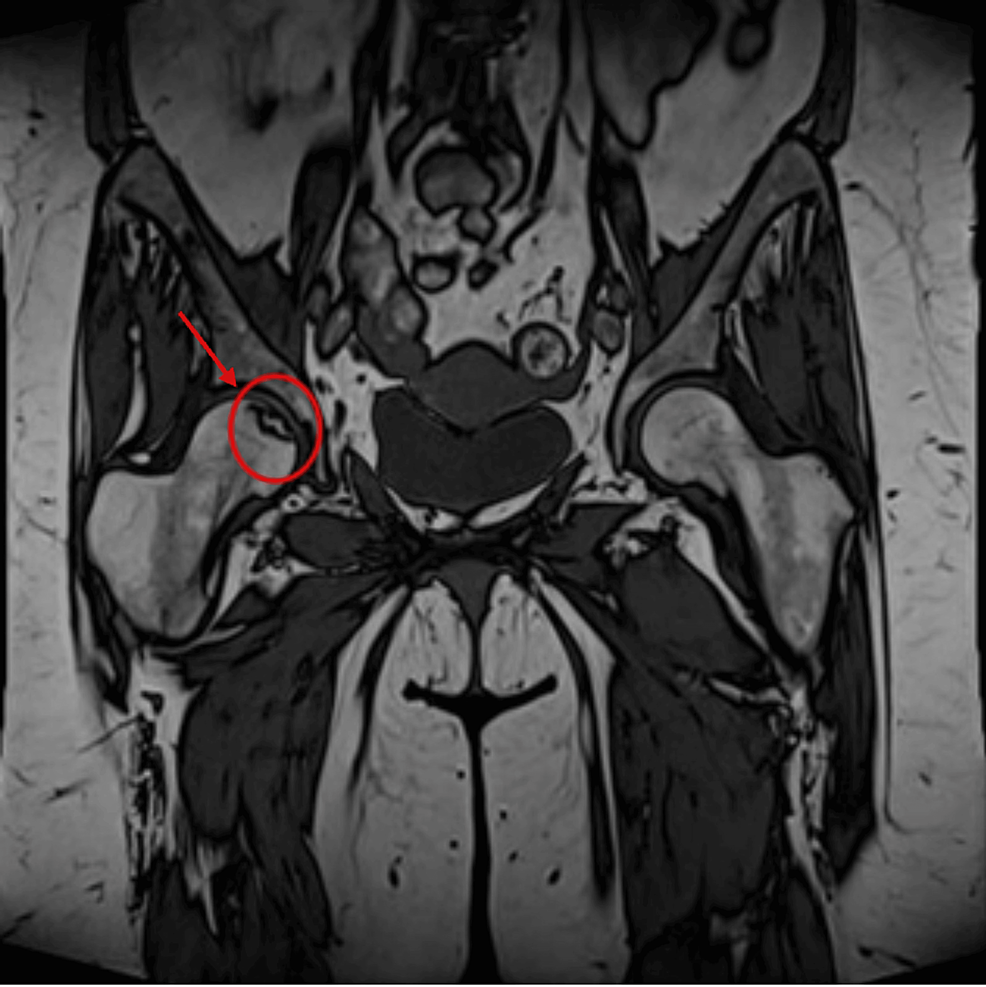
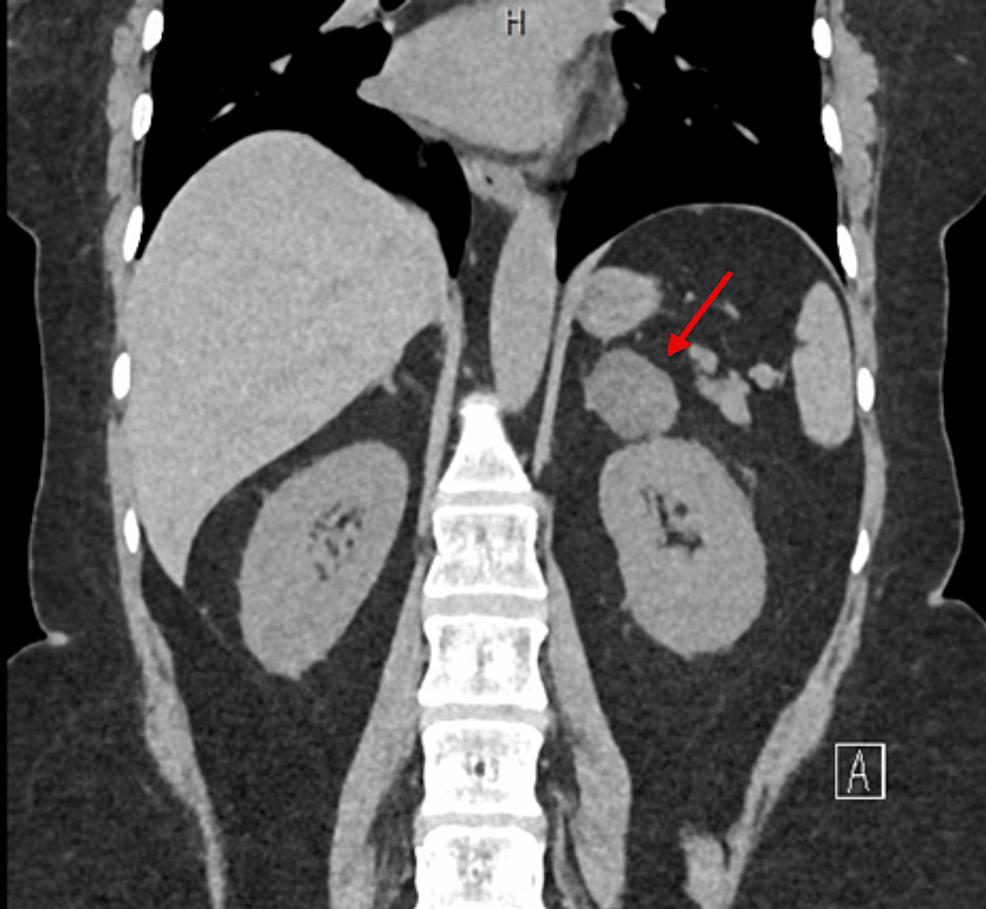
During hospitalization, she underwent further investigation for hypokalaemia and resistant hypertension, which showed an elevated 24-h urine free cortisol (24-h UFC) of 1904.4 nmol/d (upper limit of normal: 485.4 nmol/d) and consequently was referred to Endocrinology for further assessment. Repeat outpatient-based investigations after discharge from the hospital confirmed an elevated 24-h UFC of 1578.4 nmol/d, elevated AM serum cortisol of 1749.2 nmol/L (normal: 80–477.3 nmol/L), non-suppressed serum cortisol of 1238.8 nmol/L (normal response: < 50 nmol/L) after a 48-h low dose dexamethasone suppression test, and an elevated serum ACTH at 8.2 pmol/L (normal: 0.5–2.2 pmol/L). MRI of the sella as well as gallium DOTATATE PET-CT did not show any demonstrable lesion (Figs 1A, B and 2A, B). Subsequently, she underwent bilateral inferior petrosal sinus sampling (BIPSS) using 100 µg ovine CRH, which showed a post-CRH central to peripheral ACTH ratio of 3, lateralizing to the right with a ratio of 2.1. Based on these findings, a diagnosis of MRI-negative CD was made.
 View Full Size
View Full SizeCitation: Endocrinology, Diabetes & Metabolism Case Reports 2024, 3; 10.1530/EDM-24-0011
 View Full Size
View Full SizeCitation: Endocrinology, Diabetes & Metabolism Case Reports 2024, 3; 10.1530/EDM-24-0011
Treatment
While awaiting surgical opinion, the patient was started on Ketoconazole 200 mg po TID. She was unable to tolerate a larger dose; therefore, cabergoline 1 mg twice a week was added. The options of trans-sphenoidal pituitary surgery and bilateral adrenalectomy were discussed with the patient, which she declined, and decided to continue with medical therapy.
Outcome and follow-up
Medical therapy was adjusted over the next several weeks until 24-h UFC normalized and remained normal during 24 months of follow-up with the most recent being 85 nmol/d. With biochemical remission of CD, her blood pressure normalized, and she required a reduction in the dose of anti-hypertensive and anti-hyperglycaemic therapy. Her serum potassium levels also normalized. She initially regained 15 lbs but called the clinic when, despite taking medical therapy, she once again began losing weight and her serum potassium dropped to 2.7 mmol/L. Repeat serum AM cortisol was significantly elevated at 935.3 nmol/L, as was 24h UFC at 1457.3 nmol/d. Further inquiry revealed that she had been prescribed omeprazole therapy by her family physician for symptoms of reflux. Omeprazole was discontinued due to its potential effect on decreasing the efficacy of ketoconazole therapy, and her cortisol and potassium levels rapidly normalized. Since then, she has regained 50 lbs, being almost back to her baseline weight, and her mobility and strength have improved from being initially bed-bound to now mobilizing independently using a walker. Pre and post therapy values are summarized in Table 1.
| Test | Reference range | At presentation | Recent follow-up |
|---|---|---|---|
| 24-h urine cortisol (nmol/TV) | ULN=486 | 1908 | 85 |
| AM cortisol (nmol/L) | 133-537 | 2371 | 796 |
| Cortisol post-48h low DMS dose (nmol/L) | <1.8 | 44.9 | NA |
| ACTH (pmol/L) | 2.3-10.1 | 37.5 | 14 |
| Potassium (mmol/L) | 3.6-5.2 | 2.4 | 4.5 |
DMS= dexamethasone suppression; NA = Not Applicable; ULN = upper limit of normal.
Discussion
Here we report an unusual case of CD presenting with features that were initially highly suggestive of ectopic ACTH production with weight loss rather than the usual weight gain. All of the initial symptoms resolved following biochemical control of hypercortisolemia. In our review of the literature, CD associated with weight loss has previously only been reported in association with severe depression, psychosis, eating disorders, or malignancy (3, 4). For instance, a case of familial CD was reported in a child who also had an intercurrent eating disorder (anorexia), which led to weight loss despite CD (3). Weight loss due to ectopic ACTH-induced CD has also been previously reported, where weight loss was thought to be due to the underlying malignancy (4). However, our patient had well-documented pituitary ACTH-induced CD.
Chronic hypercortisolemia is associated with increased abdominal adiposity that is thought to be caused by the downregulation of adenosine monophosphate-activated protein kinase (AMPK), which is responsible for regulating lipid metabolism (5). Furthermore, glucocorticoids also induce a direct orexigenic effect, which leads to weight gain (6). Weight loss in association with hypercortisolemia, on the contrary, can be a presenting feature of ectopic ACTH-producing tumors such as small cell lung cancer. While the underlying mechanism of weight loss is not fully understood, it is thought to be partly due to cAMP/Protein kinase A (PKA) pathway activation, with an increase in PKA activity resulting in altered downstream regulation of cAMP-related lipogenic and lipolytic proteins (6). In addition, high ACTH secretion and the malignant characteristics of the neoplastic process are also thought to play roles in weight loss (7). Our patient had no evidence of ectopic ACTH production.
A previous study (8) comparing the clinical features of CD in older vs younger patients reported that weight gain was more common in younger individuals, whereas older patients typically presented with catabolic changes, likely due to age-related variability in tissue sensitivity to glucocorticoid receptors and intracellular cortisol signaling. The overall rates of central adiposity were 71.1% in older patients compared with 80.0% in younger patients (8).
Another unusual feature was hypokalemia, which is generally associated with ectopic ACTH production. However, up to 10% of CD patients present with low potassium. Hypokalemia is caused by the mineralocorticoid effect of excess cortisol. Supraphysiologic production of cortisol tends to saturate 11β-hydroxysteroid dehydrogenase type II (11β-HSD2) activity in the renal tubule, which is primarily responsible for converting active cortisol into inactive cortisone. This could lead to excess binding of cortisol to mineralocorticoid receptors, resulting in an increase in potassium excretion and thus hypokalemia. While some studies have suggested that ACTH can also lead to lowering 11β-HSD2 activity causing hypokalemia, others have not found any such correlation (9, 10). In our patient, serum potassium normalized after she achieved eucortisolemia with medical therapy.
Declaration of interest
The authors declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of the study reported.
Funding
This research did not receive any specific grant from any funding agency in the public, commercial, or not-for-profit sector.
Patient consent
Written informed consent for publication of their clinical details and/or clinical images was obtained from the patient/parent/guardian/relative of the patient.
Author contribution statement
All authors reviewed the results and approved the final version of the manuscript.
Acknowledgements
We thank Dr Brian Moses (Yarmouth Regional Hospital, NS, Canada) and Dr Scott Lee (Valley Regional Hospital, NS, Canada) for their contributions in managing the patient.
References
-
1↑Pivonello R, De Martino MC, De Leo M, Simeoli C, & Colao A. Cushing’s disease: the burden of illness. Endocrine 2017 56 10–18. (https://doi.org/10.1007/s12020-016-0984-8)
-
2↑Fleseriu M, Auchus R, Bancos I, Ben-Shlomo A, Bertherat J, Biermasz NR, Boguszewski CL, Bronstein MD, Buchfelder M, Carmichael JD, et al.Consensus on diagnosis and management of Cushing’s disease: a guideline update. Lancet Diabetes and Endocrinology 2021 9 847–875. (https://doi.org/10.1016/S2213-8587(2100235-7)
-
3↑Miller AH, & Yeung SCJ. Hypercortisolism manifesting as severe weight loss, hypokalemia, and hyperglycemia in the emergency department. Journal of Emergency Medicine 2016 50 e187–e190. (https://doi.org/10.1016/j.jemermed.2015.06.084)
-
4↑Cameron FJ, & Warne GL. Familial Cushing’s disease with severe weight loss occurring in late childhood. Journal of Paediatrics and Child Health 1997 33 74–77. (https://doi.org/10.1111/j.1440-1754.1997.tb00996.x)
-
5↑Kola B, Christ-Crain M, Lolli F, Arnaldi G, Giacchetti G, Boscaro M, Grossman AB, & Korbonits M. Changes in adenosine 5′-monophosphate-activated protein kinase as a mechanism of visceral obesity in Cushing’s syndrome. Journal of Clinical Endocrinology and Metabolism 2008 93 4969–4973. (https://doi.org/10.1210/jc.2008-1297)
-
6↑Ferrau F, & Korbonits M. Metabolic comorbidities in Cushing’s syndrome. European Journal of Endocrinology 2015 173 M133–M157. (https://doi.org/10.1530/EJE-15-0354)
-
7↑Salgado LR, Fragoso MCBV, Knoepfelmacher M, Machado MC, Domenice S, Pereira MAA, & de Mendonça BB. Ectopic ACTH syndrome: our experience with 25 cases. European Journal of Endocrinology 2006 155 725–733. (https://doi.org/10.1530/eje.1.02278)
-
8↑Qiao N, Swearingen B, & Tritos NA. Cushing’s disease in older patients: presentation and outcome. Clinical Endocrinology 2018 89 444–453. (https://doi.org/10.1111/cen.13799)
-
9↑Fan L, Zhuang Y, Wang Y, Liu X, Liu D, Xiang B, He M, Zhang Z, Li Y, Wang Y, et al.Association of hypokalemia with cortisol and ACTH levels in Cushing’s disease. Annals of the New York Academy of Sciences 2020 1463 60–66. (https://doi.org/10.1111/nyas.14205)
-
10↑Shah NZ, Malik S, Sathyapalan T, & Mohammed K. Refractory hypokalaemia and hypertension with metabolic alkalosis: an acute presentation of Cushing’s disease secondary to a pituitary macroadenoma. BMJ Case Reports 2021 14 e244850. (https://doi.org/10.1136/bcr-2021-244850)
From https://edm.bioscientifica.com/view/journals/edm/2024/3/EDM24-0011.xml
Filed under: Cushing's, symptoms | Tagged: Cushing's, hypokalemia, weight loss | Leave a comment »



 Data were derived from Rudman Y, et al. Eur J Endocrinol. 2024;doi:10.1093/ejendo/lvae098.
Data were derived from Rudman Y, et al. Eur J Endocrinol. 2024;doi:10.1093/ejendo/lvae098.