Filed under: Cushing's, pituitary, symptoms, Treatments | Tagged: 3-Tesla Dynamic Pituitary MRI, ACTH, endoscopic trans-nasal neuro-navigation, facial puffiness, headache, high blood pressure, irregular menstrual cycles, kidney stones, pituitary microadenoma, Weight gain | Leave a comment »
The Reflex Dexamethasone Suppression Test: Development and Assessment of Reflexed Serum Dexamethasone Measurement for the Diagnosis of Cushing Syndrome
Abstract
Background
Objective
Methods
Results
Conclusion
Abbreviations
Highlights
- •
Reflexing only nonsuppressed serum cortisol samples for the measurement of serum dexamethasone does not negatively affect the performance of the overnight low-dose DST (oDST)
- •
Reflex implementation greatly reduced the number of serum dexamethasone measurements thereby decreasing unnecessary costs
- •
The oDST appeared to be valid as long as there was a measurable serum dexamethasone result (>50 ng/dL)
Clinical Relevance
Introduction
Materials and Methods
Study Design
Study Population
Procedures
Data Collection
Outcomes Assessment
Statistical Analysis
Results
Study Population Characteristics

Figure 1. Flowchart of participants selection from 616 completed oDSTs completed 6 months before (n = 308) and after (n = 308) Reflex implementation. Subsequent oDSTs for the same patient and unmeasurable post-oDST serum dexamethasone (SerDex) (<50 ng/dL [Lower quantifiable limit]) were excluded from analysis. A total of 542 oDSTs were included for analysis and breakdown of CS diagnosis and etiology are shown. ACTH-dependent CS is further broken down to differentiate neoplastic (NH) versus non-neoplastic (NNH) etiologies. CS = Cushing Syndrome; NH = neoplastic hypercortisolism; NNH = nonneoplastic hypercortisolism; oDST = overnight dexamethasone suppression test; SerDex = serum dexamethasone.
Table 1. Demographic Characteristics of Patients Who Underwent oDST Before and After Reflex Implementation
| Empty Cell | Pre-Reflex | Post-Reflex | ||||
|---|---|---|---|---|---|---|
| Yes CS with NNH | Yes CS without NNH | No CS | Yes CS with NNH | Yes CS without NNH | No CS | |
| N | 38 | 34 | 223 | 40 | 38 | 241 |
| Age | ||||||
| Mean (SD) | 63.6 (13.8) | 63.8 (14.4) | 56.0 (15.1)a | 63.8 (13.2) | 63.1 (13.1) | 55.3 (15.5)b |
| Sex | ||||||
| Male (%) | 6 (15.8) | 4 (11.8) | 57 (25.6) | 11 (27.5) | 11 (28.9) | 67 (27.8) |
| Female (%) | 32 (84.2) | 30 (88.2) | 166 (74.4) | 29 (72.5) | 27 (71.1) | 174 (72.2) |
| Race | ||||||
| American Indian or Alaskan Native (%) | 0 | 0 | 1 (0.4) | 0 | 0 | 1 (0.4) |
| Asian (%) | 0 | 0 | 3 (1.3) | 0 | 0 | 1 (0.4) |
| Black or African American (%) | 5 (13.2) | 5 (14.7) | 27 (12.2) | 6 (15.0) | 6 (15.8) | 23 (9.5) |
| Other (%) | 2 (5.3) | 1 (2.9) | 8 (3.6) | 1 (2.5) | 1 (2.2) | 9 (3.8) |
| White (%) | 31 (81.5) | 28 (82.4) | 184 (82.5) | 33 (82.5) | 31 (82.0) | 207 (85.9) |
- a
-
Age different from group with CS within Pre-Reflex-oDST (P = 0.005).
- b
-
Age different from group with CS within Post-Reflex-oDST (P < 0.001) regardless of whether NNH cases are included. Male vs female distribution NS (χ2 = 2.533, 3 df, P = 0.469). Race distribution NS (χ2 = 4.37733, 12 df, P = 0.976).
Prereflex-oDST vs Post-reflex-oDST Analysis

Figure 2. Comparison of oDST serum cortisol (SerCort) levels Pre-vs Post-Reflex-oDST implementation. The medians are further stratified based on whether the patient did not have Cushing Syndrome (No CS – red) and those who had CS (Yes CS – blue). Each box represents the interquartile range and the horizontal line within represents the median. The error bars represent the 10th-90th percentiles and dots represent results outlying the 10th-90th percentiles. a, denotes significant difference of median SerCort levels between no CS vs CS in both Pre- and Post-Reflex-oDST groups (P < 0.001). There was no difference in medians following exclusion of NNH from Yes CS in both Pre- and Post-Reflex-oDST groups (P = 0.269). CS = Cushing Syndrome; NH = neoplastic hypercortisolism; NNH = nonneoplastic hypercortisolism; oDST = overnight dexamethasone suppression test; SerDex = serum dexamethasone.
Table 2. Receiver operating characteristic (ROC) analysis of oDST SerCort results for Pre-vs Post-Reflex-oDST groups. A. Analysis including NNH patients are at the top; B. Analysis excluding NNH patients are at the bottom
| Empty Cell | Pre-Reflex | Post-Reflex | ||||
|---|---|---|---|---|---|---|
| A. Including NNH patients | ||||||
| ROC Curve Area (SE) | 0.97 (0.01) | 0.97 (0.01) | ||||
| 95% confidence interval | 0.96-0.99 | 0.95-0.99 | ||||
| P value | P < 0.0001 | P < 0.0001 | ||||
| Sample size: No CS/Yes CS | 223/38 | 241/40 | ||||
| Cutoff | Sensitivity | Specificity | Cutoff | Sensitivity | Specificity | |
| Optimal 8 AM SerCort Cutoff (mcg/dL) | 2.1 | 92% | 93% | 2.1 | 95% | 93% |
| Empty Cell | Pre-Reflex without NNH | Post-Reflex without NNH | ||||
|---|---|---|---|---|---|---|
| B. Excluding NNH patients | ||||||
| ROC curve area (SE) | 0.97 (0.01) | 0.97 (0.01) | ||||
| 95% confidence interval | 0.96-0.99 | 0.95-0.99 | ||||
| P Value | P < 0.0001 | P < 0.0001 | ||||
| Sample size: No CS/Yes CS | 223/34 | 241/38 | ||||
| Cutoff | Sensitivity | Specificity | Cutoff | Sensitivity | Specificity | |
| Optimal 8 AM SerCort Cutoff (mcg/dL) | 2.1 | 91% | 92% | 2.1 | 95% | 93% |
Prereflex-oDST Comparison of SerDex vs SerCort

Figure 3. Comparison of post-oDST serum cortisol (SerCort) to serum dexamethasone (SerDex) in Pre-Reflex-oDST group. (A) Comparison of post-oDST SerCort and SerDex for no CS patients in the Pre-Reflex-oDST group. SerCort in the No CS patients stratified by the ARUP Lower limit of the reference range for SerDex (140 ng/dL). There was no significant difference in median SerCort with the SerDex <140 ng/dL (N = 20) and >140 ng/dL (N = 203) groups (1.2 vs 1.1 mcg/dL, respectively, P = 0.621). (B) Comparison of all Pre-Reflex-oDST group oDSTs stratified by patients with (blue) and without (red) CS. The black vertical solid line represents the limit of quantitation (LOQ) of SerDex (50 ng/dL). There was no correlation of SerDex and SerCort achieved in either group (see text for specifics). There was no correlation when NNH cases were removed as well (P = 0.432). CS = Cushing Syndrome; NH = neoplastic hypercortisolism; NNH = nonneoplastic hypercortisolism; oDST = overnight dexamethasone suppression test; SerDex = serum dexamethasone.
Discussion
Conclusion
Disclosure
Acknowledgment
References
- 1
H. Raff, T. CarrollCushing’s syndrome: from physiological principles to diagnosis and clinical careJ Physiol, 593 (3) (2015), pp. 493-506
- 2
J.W. Findling, H. RaffRecognition of nonneoplastic hypercortisolism in the evaluation of patients with cushing syndromeJ Endocr Soc, 7 (8) (2023), Article bvad087
- 3
J.W. Findling, H. RaffDiagnosis of endocrine disease: differentiation of pathologic/neoplastic hypercortisolism (Cushing’s syndrome) from physiologic/non-neoplastic hypercortisolism (formerly known as pseudo-cushing’s syndrome)Eur J Endocrinol, 176 (5) (2017), pp. R205-R216
- 4
M.S. Sagmeister, L. Harper, R.S. HardyCortisol excess in chronic kidney disease – a review of changes and impact on mortalityFront Endocrinol (Lausanne), 13 (2022), Article 1075809
- 5
R. Pivonello, A.M. Isidori, M.C. De Martino, J. Newell-Price, B.M. Biller, A. ColaoComplications of cushing’s syndrome: state of the artLancet Diabetes Endocrinol, 4 (7) (2016), pp. 611-629
- 6
M. Araujo-Castro, E. Pascual-Corrales, C. LamasPossible, probable, and certain hypercortisolism: a continuum in the risk of comorbidityAnn Endocrinol (Paris), 84 (2) (2023), pp. 272-284
- 7
M. Fassnacht, W. Arlt, I. Bancos, et al.Management of adrenal incidentalomas: European society of endocrinology clinical practice guideline in collaboration with the european network for the study of adrenal tumorsEur J Endocrinol, 175 (2) (2016), pp. G1-G34
- 8
M. Fleseriu, R. Auchus, I. Bancos, et al.Consensus on diagnosis and management of Cushing’s disease: a guideline updateLancet Diabetes Endocrinol, 9 (12) (2021), pp. 847-875
- 9
P.J. Wood, J.H. Barth, D.B. Freedman, L. Perry, B. SheridanEvidence for the low dose dexamethasone suppression test to screen for Cushing’s syndrome–recommendations for a protocol for biochemistry laboratoriesAnn Clin Biochem, 34 (Pt 3) (1997), pp. 222-229
- 10
L.K. Nieman, B.M. Biller, J.W. Findling, et al.The diagnosis of cushing’s syndrome: an endocrine society clinical practice guidelineJ Clin Endocrinol Metab, 93 (5) (2008), pp. 1526-1540
- 11
H. Raff, S.T. Sharma, L.K. NiemanPhysiological basis for the etiology, diagnosis, and treatment of adrenal disorders: cushing’s syndrome, adrenal insufficiency, and congenital adrenal hyperplasiaCompr Physiol, 4 (2) (2014), pp. 739-769
- 12
R.S. Mohamed, B. Abuelgasim, S. Barker, et al.Late-night salivary cortisol and cortisone should be the initial screening test for cushing’s syndromeEndocr Connect, 11 (7) (2022), Article e220050
- 13
S. GuthrieThe impact of dexamethasone pharmacokinetics on the DST: a reviewPsychopharmacol Bull, 27 (4) (1991), pp. 565-576
- 14
T. Nickelsen, W. Lissner, K. SchofflingThe dexamethasone suppression test and long-term contraceptive treatment: measurement of ACTH or salivary cortisol does not improve the reliability of the testExp Clin Endocrinol, 94 (3) (1989), pp. 275-280
- 15
A.W. MeikleDexamethasone suppression tests: usefulness of simultaneous measurement of plasma cortisol and dexamethasoneClin Endocrinol (Oxf), 16 (4) (1982), pp. 401-408
- 16
M. Fleseriu, A.H. Hamrahian, A.R. Hoffman, et al.American association of clinical endocrinologists and American college of endocrinology disease state clinical review: diagnosis of recurrence in cushing diseaseEndocr Pract, 22 (12) (2016), pp. 1436-1448
- 17
G.A. Ueland, P. Methlie, R. Kellmann, et al.Simultaneous assay of cortisol and dexamethasone improved diagnostic accuracy of the dexamethasone suppression testEur J Endocrinol, 176 (6) (2017), pp. 705-713
- 18
N. Genere, R.J. Kaur, S. Athimulam, et al.Interpretation of abnormal dexamethasone suppression test is enhanced with use of synchronous free cortisol assessmentJ Clin Endocrinol Metab, 107 (3) (2022), pp. e1221-e1230
- 19
B.R. Javorsky, H. Raff, T.B. Carroll, et al.New cutoffs for the biochemical diagnosis of adrenal insufficiency after ACTH stimulation using specific cortisol assaysJ Endocr Soc, 5 (4) (2021), Article bvab022
- 20
Centers for medicare & medicaid services – clinical laboratory fee scheduleAvailable from:https://www.cms.gov/medicare/payment/fee-schedules/clinical-laboratory-fee-schedule-clfs, Accessed 31st Jan 2025
- 21
A.H. Rosenbaum, A.F. Schatzberg, R.A. MacLaughlin, et al.The dexamethasone suppression test in normal control subjects: comparison of two assays and effect of ageAm J Psychiatry, 141 (12) (1984), pp. 1550-1555
- 22
S.M. RoperYield of serum dexamethasone measurement for reducing false-positive results of low-dose dexamethasone suppression testingJ Appl Lab Med, 6 (2) (2021), pp. 480-485
- 23
N. Vogg, M. Kurlbaum, T. Deutschbein, B. Grasl, M. Fassnacht, M. KroissMethod-specific cortisol and dexamethasone thresholds increase clinical specificity of the dexamethasone suppression test for cushing syndromeClin Chem, 67 (7) (2021), pp. 998-1007
- 24
D.G. Farinelli, K.C. Oliveira, L.F. Hayashi, C.E. KaterOvernight 1-mg dexamethasone suppression test for screening cushing syndrome and mild autonomous cortisol secretion (MACS): what happens when serum dexamethasone is below cutoff? How frequent is it?Endocr Pract, 29 (12) (2023), pp. 986-993
- 25
F. Ceccato, C. Artusi, M. Barbot, et al.Dexamethasone measurement during low-dose suppression test for suspected hypercortisolism: threshold development with and validationJ Endocrinol Invest, 43 (8) (2020), pp. 1105-1113
- 26
M. Keller-WoodHypothalamic-Pituitary–Adrenal axis-feedback controlCompr Physiol, 5 (3) (2015), pp. 1161-1182
- 27
E. Valassi, B. Swearingen, H. Lee, et al.Concomitant medication use can confound interpretation of the combined dexamethasone-corticotropin releasing hormone test in cushing’s syndromeJ Clin Endocrinol Metab, 94 (12) (2009), pp. 4851-4859
- 28
C.W. Wilkinson, E.R. Peskind, M.A. RaskindDecreased hypothalamic-pituitary-adrenal axis sensitivity to cortisol feedback inhibition in human agingNeuroendocrinology, 65 (1) (1997), pp. 79-90
Filed under: Cushing's, Diagnostic Testing | Tagged: ACTH, dexamethasone suppression test, endogenous, low-dose overnight serum dexamethasone suppression test, ODST | Leave a comment »
A Case 0f Hailey–Hailey Disease Accompanied by Cushing’s Syndrome and Adrenal Insufficiency Due to Long-Term Usage of Topical Steroids With Review of Literature
Hailey–Hailey disease (HHD), or familial benign chronic pemphigus, is a rare autosomal dominant disorder characterized by recurrent vesicles and erosions in intertriginous areas. Topical corticosteroids are the primary treatment, but their potential systemic side effects are often overlooked. Prolonged use on compromised skin can lead to excessive absorption, increasing the risk of iatrogenic Cushing’s syndrome and adrenal insufficiency.
Here, we report the case of a 50-year-old woman with HHD who had been using topical clobetasol or betamethasone for over 10 years, reaching doses up to 50 g/day.
She developed Cushingoid features, metabolic abnormalities, and suppression of the hypothalamic–pituitary–adrenal (HPA) axis. After tapering off topical corticosteroids, she developed adrenal insufficiency and associated withdrawal symptoms. Following the initiation of hydrocortisone replacement therapy, psychiatric symptoms, impaired glucose tolerance, and osteoporotic fractures emerged, suggesting exacerbation of iatrogenic Cushing’s syndrome.
This case highlights the risk of systemic complications from chronic topical corticosteroid use, particularly in high-absorption areas. Gradual dose reduction, close endocrine monitoring, and individualized tapering strategies are essential to prevent severe outcomes.
Clinicians should be aware of potential adrenal suppression and consider endocrine evaluation in patients receiving prolonged, high-dose topical corticosteroid therapy.
Filed under: adrenal, Cushing's, Rare Diseases | Tagged: adrenal insufficiency, Cushing’s syndrome, familial benign chronic pemphigus, Familial chronic benign pemphigus, Hailey-Hailey Disease, steroid, Steroid withdrawal symptom | Leave a comment »
Cushing’s Syndrome in a Young Woman Due to Prolonged Betamethasone Nasal Drop Use
Abstract
Background
Cushing’s syndrome is an uncommon but serious condition caused by long-term exposure to elevated cortisol levels, which is usually iatrogenic in origin. Although systemic corticosteroids are the most frequent agents, the association of intranasal corticosteroids with this condition is remarkably rare.
Case presentation
This report is about a 21-year-old Iranian woman using betamethasone nasal drops for nasal obstruction. The patient presented with weight gain, Amenorrhea, mood disturbances, red purplish striae, and mild hirsutism. Hormonal assessments revealed suppression of the hypothalamic–pituitary–adrenal axis.
Conclusion
This case demonstrates the underappreciated systemic effects of intranasal betamethasone to induce Cushing’s syndrome. It serves as a pivotal reminder of the need for vigilance in prescribing practices and reinforces the importance of early diagnosis to ensure favorable patient outcomes.
Background
Iatrogenic Cushing’s syndrome (CS) is an endocrine disease caused by long-term or high-dose glucocorticoid use [1]. Although iatrogenic cases are commonly associated with oral or injectable glucocorticoids [2], few reports described CS after the use of intranasal steroid sprays (INS) such as betamethasone in adults [3,4,5,6,7]. Currently, INS is widely used for managing conditions such as allergic rhinitis, nasal polyposis, and other upper airway disorders owing to their localized effects and limited systemic absorption [8, 9]. However, prolonged use, high doses, or using potent formulations can lead to significant systemic absorption, resulting in Hypothalamic–pituitary–adrenal (HPA) axis suppression, and frank CS [10]. Betamethasone nasal spray, a cornerstone in the treatment of nasal congestion, has the potential for systemic absorption by the nasal mucosa, particularly with prolonged or excessive use [11].
This report presents the case of a young woman who developed CS following the overuse of betamethasone nasal drops. It also highlights the importance of detailed patient histories when diagnosing CS and highlights the critical need to educate patients on the proper use and potential risks of steroid therapies to prevent complications. This case report adheres to the case report (CARE) guidelines [12].
Case presentation
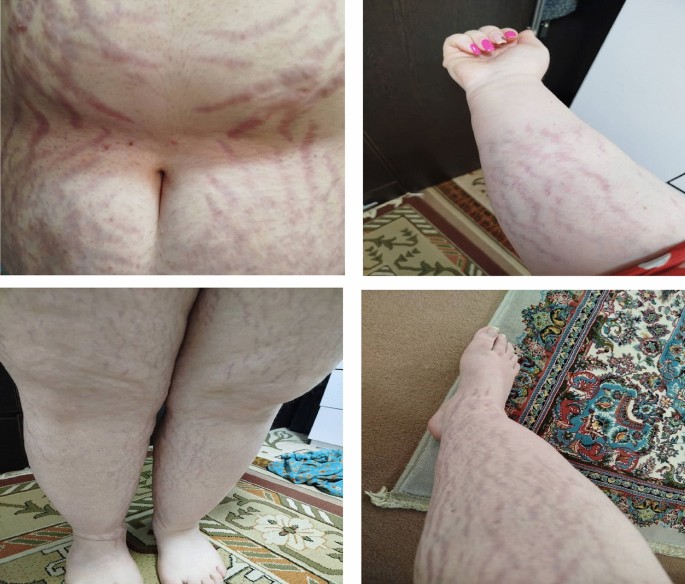
This is the case of a 21-year-old Iranian female who presented with a history of rapid weight gain (30 kg in 8 months), irregular menstrual cycles, and significant mood changes. Her body mass index (BMI) was calculated at 40.07 kg/m2, classifying her as obese, and her blood pressure was recorded at 115/75 mmHg. In addition, she exhibited red–purple striae on her abdomen and limbs and mild hirsutism (modified Ferriman–Gallwey Score (FGS) score = 10), prompting admission for further evaluation after multiple outpatient visits yielded no definitive diagnosis.
Figure 1 is a clinical photograph (with patient consent) or an illustration of the red–purple striae.
Clinical photograph showcasing the red–purplish striae on the patient’s abdomen, arms, and lower limbs
Upon admission, the patient’s history revealed prolonged use of betamethasone 0.1% 1 mg/mL nasal drops, administered at a daily dosage of 5 cc, in combination with oxymetazoline (a sympathomimetic nasal preparation) at a daily dosage of 1 cc, over approximately 12 months, to address nasal obstruction. Her symptoms began 6 months after starting the nasal drops. Further medication history revealed no other corticosteroid use. Notably, the patient had a past diagnosis of polycystic ovary (PCO) syndrome made on the basis of Rotterdam 2003 criteria (oligomenorrhea since menarche and clinically androgen excess) but did not undergo treatment or maintain laboratory records.
A detailed hormonal evaluation was undertaken. Morning plasma cortisol less than 0.05 µg/dL and adrenocorticotropic hormone (ACTH) less than 5 (10–56 pg/mL) measurements were abnormally low. Her 24-hour urine-free cortisol concentrations of 1.04 µg/24 h were significantly reduced, indicating suppression of the HPA axis secondary to prolonged exogenous corticosteroid exposure. All tests were repeated several times by endocrinologists during the time course of disease manifestations.
Table 1 summarizes the hormonal test results to clearly display the abnormalities.
Imaging studies before admission included a computed tomography (CT) scan of the adrenal glands, which showed that both adrenal glands were of normal size. However, a dynamic pituitary magnetic resonance imaging (MRI) revealed an 11 mm pituitary gland, despite there being no rationale for imaging studies in this scenario.
The patient was counseled extensively about the condition, and betamethasone nasal drops were discontinued immediately. Ear, nose, and throat (ENT) consultation revealed normal findings and the psychiatric team diagnosed her with major depressive disorder (MDD). She was discharged on 15 mg prednisolone with a structured tapering plan to allow for gradual recovery of adrenal function and to prevent acute adrenal insufficiency. Follow-up appointments were scheduled to monitor her clinical progress and re-evaluate her HPA axis recovery.
Discussion
This case highlights the rare but significant occurrence of iatrogenic CS secondary to prolonged use of intranasal betamethasone. Although oral corticosteroids are well-known to cause HPA axis suppression, INS is generally considered safer owing to their localized effects and lowering systemic absorption side effects. However, the associated potential of systemic absorption in INS remains a concern [13]. As demonstrated in this case, prolonged use of potent formulations such as betamethasone can lead to significant systemic effects, particularly when administered inappropriately or at high doses.
Betamethasone nasal drops, although effective for treating nasal congestion and inflammation [14, 15], carry a potential risk of systemic absorption through the nasal mucosa. Factors, such as prolonged use [6, 16, 17], and high potency [18], can significantly increase systemic bioavailability. R. J. Perry et al. [19] in study of seven children highlights that even patients receiving doses within conventional safety ranges may exhibit varying sensitivity to glucocorticoids, leading to symptomatic adrenal suppression or glucocorticoid excess. Unlike newer corticosteroid compounds, such as fluticasone or mometasone, which undergo extensive first-pass metabolism in the liver, betamethasone exhibits minimal hepatic metabolism, contributing to its prolonged systemic activity [20, 21]. This pharmacokinetic profile underscores the need for careful regulation and monitoring of its use, even in ostensibly localized therapies.
The clinical manifestations in this patient, including central obesity, striae, hirsutism, and mood changes, were classic features of CS and guided the diagnostic process [22]. Scutelnicu et al. [23] reported a case of a patient in the second trimester of pregnancy who, owing to chronic sinusitis, underwent intranasal betamethasone spray therapy. The patient manifested extensive striae on the lower limbs, as well as edema in the legs, arms, and face, accompanied by a weight gain of 22 kg over 3 months. After switching the patient’s treatment to an alpha-1 adrenergic agonist spray, the condition was managed uneventfully without any symptoms of adrenal insufficiency.
Requesting imaging assessments, including a CT scan and MRI, as a first step further complicated the diagnostic process. This highlights a common diagnostic pitfall: the use of imaging as an initial approach can lead to the discovery of incidentalomas, which may misdirect clinical attention. Such findings risk overshadowing the primary etiology of the condition, potentially resulting in misdiagnosis or delayed treatment. This emphasizes the importance of prioritizing functional assessments over imaging in the early diagnostic workup to avoid unwarranted diagnostic confusion and ensure accurate identification of the underlying pathology.
Management involved the immediate cessation of betamethasone nasal drops and initiation of a structured tapering regimen with prednisolone to support adrenal recovery. The importance of stress-dose precautions during intercurrent illnesses was emphasized, alongside comprehensive patient education to prevent future misuse of corticosteroids. The gradual improvement in adrenal function during follow-up highlights the reversibility of glucocorticoid-induced adrenal suppression with appropriate intervention.
Conclusion
This case underscores several critical lessons. First, it emphasizes the importance of heightened awareness among healthcare providers regarding the potential systemic effects of topical corticosteroids, particularly potent formulations such as betamethasone. Second, it highlights the need for thorough history-taking and detailed patient education to prevent corticosteroid misuse. This report contributes to the limited body of literature on iatrogenic CS from intranasal corticosteroids, particularly in adults. Documenting the clinical presentation, diagnostic challenges, and successful management of this case, provides valuable insights into preventing, recognizing, and treating similar cases. It serves as a reminder of the delicate balance between therapeutic benefit and potential harm in corticosteroid therapy and advocates for ongoing research to establish safer prescribing practices.
Data availability
The data analyzed and generated in this study can be accessed through the corresponding author upon reasonable request.
Abbreviations
- CS:
- Cushing’s syndrome
- INS:
- Intranasal corticosteroids
- HPA axis:
- Hypothalamic–pituitary–adrenal axis
- BMI:
- Body mass index
- FGS:
- Ferriman–Gallwey Score
- PCO:
- Polycystic ovary
- ACTH:
- Adrenocorticotropic hormone
- CT:
- Computed tomography
- MRI:
- Magnetic resonance imaging
- ENT:
- Ear, nose, and throat
- MDD:
- Major depressive disorder
References
-
Cristante J, Chabre O. Factitious, or iatrogenic but unexpected Cushing’s syndrome. Ann Endocrinol (Paris). 2023;84(3):370–2.
-
Lacroix A, Feelders RA, Stratakis CA, Nieman LK. Cushing’s syndrome. Lancet. 2015;386(9996):913–27.
-
Scutelnicu A, Panaitescu AM, Ciobanu AM, Gica N, Botezatu R, Peltecu G, et al. Iatrogenic cushing’s syndrome as a consequence of nasal use of betamethasone spray during pregnancy. Acta Endocrinol. 2020;16(4):511–7.
-
Sakamoto M, Morita K, Okamura E, Uchino T, Okamoto K, Ozawa Y, et al. A case of iatrogenic cushing syndrome due to overuse of nasal steriod and concurrent administration of clarithromycin. Teikyo Med J. 2018;41(4):161–8.
-
Nutting CM, Page SR. Iatrogenic Cushing’s syndrome due to nasal betamethasone: a problem not to be sniffed at! Postgrad Med J. 1995;71(834):231–2.
-
Stevens DJ. Cushing’s syndrome due to the abuse of betamethasone nasal drops. J Laryngol Otol. 1988;102(3):219–21.
-
Dow A, Yu R, Carmichael J. Too little or too much corticosteroid? Coexisting adrenal insufficiency and Cushing’s syndrome from chronic, intermittent use of intranasal betamethasone. Endocrinol Diabetes Metab Case Rep. 2013;2013:13–0036.
-
Bachert C, Desrosiers MY, Hellings PW, Laidlaw TM. The role of biologics in chronic rhinosinusitis with nasal polyps. J Allergy Clin Immunol Pract. 2021;9(3):1099–106.
-
Bachert C, Han JK, Wagenmann M, Hosemann W, Lee SE, Backer V, et al. EUFOREA expert board meeting on uncontrolled severe chronic rhinosinusitis with nasal polyps (CRSwNP) and biologics: definitions and management. J Allergy Clin Immunol. 2021;147(1):29–36.
-
Quddusi S, Browne P, Toivola B, Hirsch IB. Cushing syndrome due to surreptitious glucocorticoid administration. Arch Intern Med. 1998;158(3):294–6.
-
Grayson JW, Harvey RJ. Topical corticosteroid irrigations in chronic rhinosinusitis. Int Forum Allergy Rhinol. 2019;9(S1):S9–15.
-
Gagnier JJ, Kienle G, Altman DG, Moher D, Sox H, Riley D. The CARE guidelines: consensus-based clinical case reporting guideline development. Global Adv Health Med. 2013;2(5):38–43.
-
McDonnell J, Weller K, Pien LC. Safety of intranasal steroids: an updated perspective. Curr Allergy Asthma Rep. 2020;20(11):69.
-
Scadding GK. Other anti-inflammatory uses of intranasal corticosteroids in upper respiratory inflammatory diseases. Allergy. 2000;55(s62):19–23.
-
Chong LY, Head K, Hopkins C, Philpott C, Burton MJ, Schilder AG. Different types of intranasal steroids for chronic rhinosinusitis. Cochrane Database Syst Rev. 2016;4(4):Cd011993.
-
Findlay CA, Macdonald JF, Wallace AM, Geddes N, Donaldson MD. Childhood Cushing’s syndrome induced by betamethasone nose drops, and repeat prescriptions. BMJ. 1998;317(7160):739–40.
-
Reynolds C, Agrawal P, McCann A, O’Sullivan T, Chroinin MN, O’Riordan SMP. Cushing syndrome and adrenal insufficiency induced by high dose prolonged intranasal betamethasone. Arch Dis Child. 2019;104:A275.
-
Oluwayemi IO, Oduwole AO, Oyenusi E, Onyiriuka AN, Abdullahi M, Fakeye-Udeogu OB, et al. Iatrogenic Cushing’s syndrome in children following nasal steroid. Pan Afr Med J. 2014;17:237.
-
Perry RJ, Findlay CA, Donaldson MDC. Cushing’s syndrome, growth impairment, and occult adrenal suppression associated with intranasal steroids. Arch Dis Child. 2002;87(1):45–8.
-
Matera MG, Rinaldi B, Calzetta L, Rogliani P, Cazzola M. Pharmacokinetics and pharmacodynamics of inhaled corticosteroids for asthma treatment. Pulm Pharmacol Ther. 2019;58: 101828.
-
Krzyzanski W, Milad MA, Jobe AH, Peppard T, Bies RR, Jusko WJ. Population pharmacokinetic modeling of intramuscular and oral dexamethasone and betamethasone in Indian women. J Pharmacokinet Pharmacodyn. 2021;48(2):261–72.
-
Savas M, Mehta S, Agrawal N, van Rossum EFC, Feelders RA. Approach to the patient: diagnosis of Cushing syndrome. J Clin Endocrinol Metab. 2022;107(11):3162–74.
-
Scutelnicu A, Panaitescu AM, Ciobanu AM, Gica N, Botezatu R, Peltecu G, et al. Iatrogenic Cushing’s syndrome as a consequence of nasal use of betamethasone spray during pregnancy. Acta Endocrinol (Buchar). 2020;16(4):511–7.
Acknowledgements
Not applicable.
Funding
Not Applicable.
Ethics declarations
Ethics approval and consent to participate
This study was conducted in accordance with ethical guidelines and was approved by the Research Ethics Committee of Iran University of Medical Sciences under approval number IR.IUMS.REC.1404.208.
Consent for publication
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
From https://jmedicalcasereports.biomedcentral.com/articles/10.1186/s13256-025-05428-3
Filed under: adrenal crisis, Cushing's, Rare Diseases | Tagged: amenorrhea, betamethasone, corticosteroids, hirsutism, iatrogenic, nasal drops, steroids, straie, stretch marks, Weight gain | Leave a comment »
Cushing Syndrome Leaves Lasting Health Effects
TOPLINE:
Compared with a matched population-based control group, patients with Cushing syndrome continued to exhibit elevated systolic and diastolic blood pressures along with reduced kidney function at least 14 years after biochemical remission.
METHODOLOGY:
- Researchers in Germany conducted a retrospective cohort study to assess the long-term trajectory of blood pressure and kidney function in patients who achieved remission of Cushing syndrome.
- They included 81 patients with Cushing syndrome (median age at baseline, 44 years; 75.3% women) and compared them with 243 matched control individuals from a population-based cohort.
- Data were collected before treatment at baseline and at median follow-up intervals of 7.1 and 14 years after biochemical remission, with assessments of blood pressure, glomerular filtration rate, the prevalence of chronic kidney disease, and the use of antihypertensives.
TAKEAWAY:
- Patients with Cushing syndrome had a significant reduction in blood pressure and required fewer antihypertensives at both 7 and 14 years vs baseline.
- However, when compared with the control group, patients with Cushing syndrome had significantly elevated systolic and diastolic pressures at baseline and 7 and 14 years post-remission (P ≤ .0002 for all).
- Although the proportion of patients on antihypertensive medications decreased in the Cushing syndrome group after remission was achieved, the prevalence of uncontrolled hypertension remained higher than in the control group at all follow-up points. In fact, reducing the use of these medications was associated with an increased risk for uncontrolled hypertension.
- Kidney function assessed via glomerular filtration rate remained consistently lower among patients with Cushing syndrome than among control individuals at baseline and 7 and 14 years post-remission (P = .005, P < .0001, and P = .0359, respectively).
IN PRACTICE:
“Our findings provide further evidence that cardiovascular effects of hypercortisolism are not entirely reversible with the normalization of cortisol levels and enhance our understanding of the deteriorative long-term cardiovascular consequences of chronic hypercortisolism,” the authors wrote.
SOURCE:
This study was led by Katrin Ritzel, Ludwig-Maximilians-Universität München (LMU Munich), LMU University Hospital in Munich, Germany. It was published online on July 29, 2025, in Journal of Endocrinological Investigation.
LIMITATIONS:
The retrospective design and single-centre nature of this study could have been considered limitations.
DISCLOSURES:
This study was supported by Else Kröner-Fresenius Stiftung. Some authors reported being supported by Deutsche Forschungsgemeinschaft, the Munich Clinician Scientist Program, the Clinician Scientist Programme on Rare Important Syndromes in Endocrinology, and other sources. All authors reported having no conflicts of interest.
https://www.medscape.com/viewarticle/cushing-syndrome-leaves-lasting-health-effects-2025a1000kj0
Filed under: Cushing's, symptoms | Tagged: blood pressure, kidney function, Post Traumatic Stress Disorder, remission | Leave a comment »