The Endocrine Society today issued a Clinical Practice Guideline (CPG) on strategies for treating Cushing’s syndrome, a condition caused by overexposure to the hormone cortisol.
The CPG, entitled “Treatment of Cushing’s Syndrome: An Endocrine Society Clinical Practice Guideline,” was published online and will appear in the August 2015 print issue of the Journal of Clinical Endocrinology and Metabolism (JCEM), a publication of the Endocrine Society.
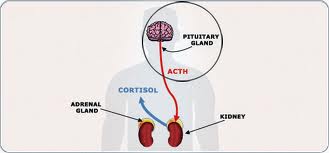
Cushing’s syndrome occurs when a person has excess cortisol in the blood for an extended period, according to the Hormone Health Network. When it is present in normal amounts, cortisol is involved in the body’s response to stress, maintains blood pressure and cardiovascular function, keeps the immune system in check, and converts fat, carbohydrates and proteins into energy. Chronic overexposure to the hormone can contribute to the development of cardiovascular disease, infections and blood clots in veins.
People who take cortisol-like medications such as prednisone to treat inflammatory conditions, including asthma and rheumatoid arthritis, can develop Cushing’s syndrome. The high cortisol levels return to normal when they stop taking the medication. This is called exogenous Cushing’s syndrome.
In other cases, tumors found on the adrenal or pituitary glands or elsewhere in the body cause the overproduction of cortisol and lead to the development of Cushing’s syndrome. The Clinical Practice Guidelines focus on this form of the condition, known as endogenous Cushing’s syndrome.
“People who have active Cushing’s syndrome face a greater risk of death – anywhere from nearly twice as high to nearly five times higher – than the general population,” said Lynnette K. Nieman, MD, of the National Institutes of Health’s Eunice Kennedy Shriver National Institute of Child Health and Human Development in Bethesda, MD, and chair of the task force that authored the guideline. “To reduce the risk of fatal cardiovascular disease, infections or blood clots, it is critical to identify the cause of the Cushing’s syndrome and restore cortisol levels to the normal range.”
In the CPG, the Endocrine Society recommends that the first-line treatment for endogenous Cushing’s syndrome be the removal of the tumor unless surgery is not possible or unlikely to address the excess cortisol. Surgical removal of the tumor is optimal because it leaves intact the hypothalamic-pituitary-adrenal axis, which is integral to the body’s central stress response.
Other recommendations from the CPG include:
- Tumors should be removed by experienced surgeons in the following situations:— A tumor has formed on one or both of the two adrenal glands.— A tumor that secretes adrenocorticotropic hormone (ACTH) – the hormone that signals the adrenal glands to produce cortisol – has formed somewhere in the body other than the adrenal or pituitary gland.
— A tumor has formed on the pituitary gland itself.
- Patients who continue to have high levels of cortisol in the blood after surgery should undergo additional treatment.
- People who had an ACTH-producing tumor should be screened regularly for the rest of their lives for high cortisol levels to spot recurrences.
- If patients’ cortisol levels are too low following surgery, they should receive glucocorticoid replacement medications and be educated about adrenal insufficiency, a condition where the adrenal glands produce too little cortisol. This condition often resolves in 1-2 years.
- Morning cortisol and/or ACTH stimulation tests, or insulin-induced hypoglycemia, can be used to test for the recovery of the hypothalamic-pituitary-adrenal axis in people who have low cortisol levels after surgery. Once the tests results return to normal, glucocorticoid replacement can be stopped.
- People who have undergone pituitary surgery should be re-evaluated for other pituitary hormone deficiencies during the post-operative period.
- Patients who have a pituitary tumor and have undergone surgery to remove both adrenal glands should be regularly evaluated for tumor progression using pituitary MRIs and tests for ACTH levels.
- Radiation therapy may be used to treat a pituitary tumor, especially if it is growing. While awaiting the effect of radiation, which may take months to years, treatment with medication is advised.
- To assess the effect of radiation therapy, the patient’s cortisol levels should be measured at 6- to 12-month intervals.
- Medications may be used to control cortisol levels as a second-line treatment after surgery for a pituitary gland tumor, as a primary treatment for ACTH-secreting tumors that have spread to other parts of the body or suspected ACTH-secreting tumors that cannot be detected on scans. Medications also can be used as adjunctive treatment to reduce cortisol levels in people with adrenal cortical carcinoma, a rare condition where a cancerous growth develops in the adrenal gland.
- People with Cushing’s syndrome should be treated for conditions associated with the disease, such as cardiovascular disease risk factors, osteoporosis and psychiatric symptoms.
- Patients should be tested for recurrence throughout their lives except in cases where the person had a benign adrenal tumor removed.
- Patients should undergo urgent treatment within 24 to 72 hours of detecting excess cortisol if life-threatening complications such as serious infection, pulmonary thromboembolism, cardiovascular complications and acute psychosis are present.
More information: The Hormone Health Network offers resources on Cushing’s syndrome at www.hormone.org/questions-and-answers/2012/cushing-syndrome
Filed under: adrenal, Cushing's, pituitary, Treatments | Tagged: ACTH, adrenal, blood pressure, cardiovascular, cortisol, Cushing's Syndrome, Dr. Lynnette Nieman, National Institute of Health, NIH, pituitary, prednisone, stress, tumor | Leave a comment »