Abstract
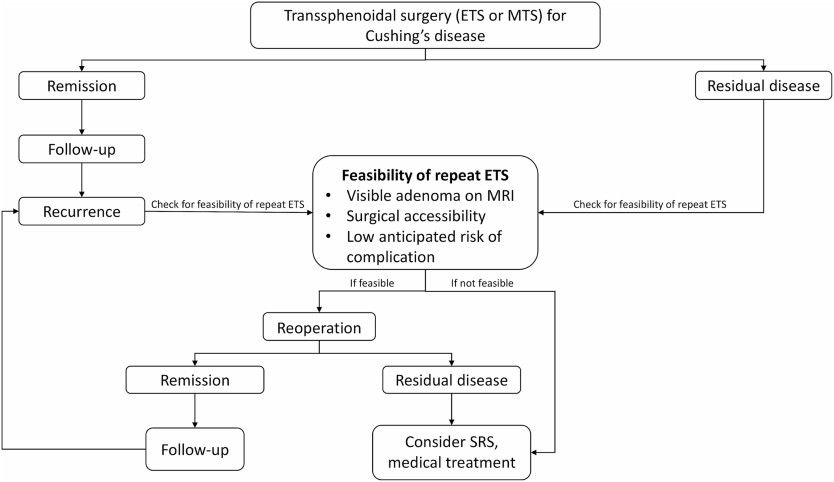
Purpose
Methods
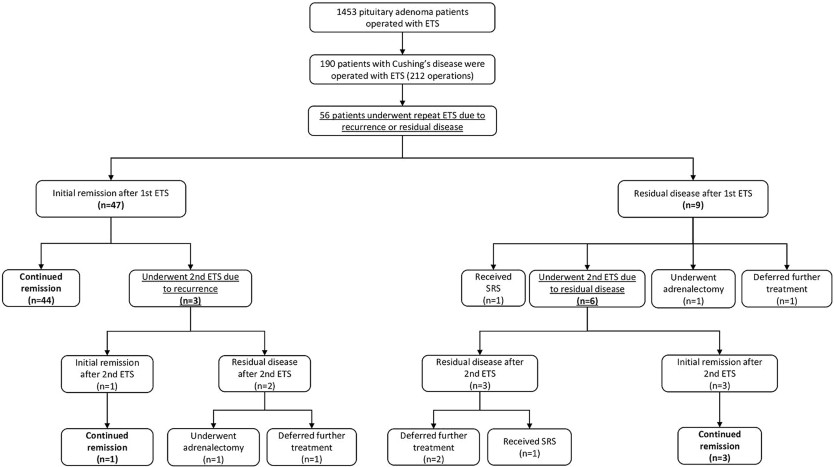
Results
Conclusion
Introduction
Methods
Study design
Patient selection
Diagnostic criteria
Routine follow-up protocol
Data collection
Statistical analysis
Results
Baseline characteristics
Hormonal data
|
variable
|
Total ( n = 65)
|
Residual disease ( n = 47)
|
Recurrence ( n = 18)
|
p -value
|
|---|---|---|---|---|
|
Technique of the previous surgery
|
< 0.001
|
|||
|
MTS
|
40 (61.5)
|
36 (76.6)
|
4 (22.2)
|
|
|
ETS
|
25 (38.5)
|
11 (23.4)
|
14 (77.8)
|
|
|
Tumor size
|
||||
|
Microadenoma
|
41 (63.1)
|
30 (63.8)
|
11 (61.1)
|
0.839
|
|
Macroadenoma
|
24 (36.9)
|
17 (36.2)
|
7 (38.9)
|
|
|
Multifocality
|
||||
|
Unifocal
|
50 (76.9)
|
37 (78.7)
|
13 (72.2)
|
0.743
|
|
Bifocal
|
15 (23.1)
|
10 (21.3)
|
5 (27.8)
|
|
|
Relation to cavernous sinus
|
||||
|
Extension
|
21 (32.3)
|
15 (31.9)
|
6 (33.3)
|
0.589
|
|
invasion
|
10 (15.4)
|
6 (12.8)
|
4 (22.2)
|
|
|
No relationship
|
34 (52.3)
|
26 (55.3)
|
8 (44.4)
|
|
|
Hardy-Wilson Classification
|
0.339
|
|||
|
Degrees
|
||||
|
I
|
38 (58.5)
|
25 (59.5)
|
8 (57.1)
|
|
|
II
|
16 (24.6)
|
8 (19)
|
5 (5)
|
|
|
III
|
6 (9.2)
|
6 (14.3)
|
1 (7.1)
|
|
|
IV
|
5 (7.7)
|
3 (7.1)
|
0 (0)
|
|
|
stage
|
0.443
|
|||
|
A
|
30 (46.2)
|
19 (45.2)
|
7 (50)
|
|
|
b
|
7 (10.8)
|
4 (9.5)
|
3 (21.4)
|
|
|
C
|
2 (3.1)
|
2 (4.8)
|
0 (0)
|
|
|
D
|
1 (1.5)
|
0 (0)
|
0 (0)
|
|
|
E
|
25 (38.5)
|
17 (40.5)
|
4 (28.6)
|
|
|
Laboratory values
|
||||
|
Preoperative serum ACTH (pg/mL)
|
182.71 ± 577.08
60.5 [37.15–104.5]
|
220.7 ± 675.73
|
83.5 ± 61.7
|
0.395
|
|
Preoperative serum cortisol (µg/dL)
|
18.75 ± 11.16
17 [12-24.65]
|
19.18 ± 12.11
|
17.64 ± 8.39
|
0.621
|
|
Postoperative serum ACTH (pg/mL)
|
43.29 ± 50.2
25.5 [15.8–53.7]
|
43.07 ± 45.42
|
43.94 ± 63.96
|
0.953
|
|
Postoperative serum cortisol (µg/dL)
|
13.41 ± 13.85
6.45 [1.77–21.01]
|
14.62 ± 14.52
|
10.25 ± 11.7
|
0.259
|
|
POD 1 Cortisol levels
|
0.700
|
|||
|
>5 µg/dL
|
35 (53.8)
|
26 (55.3)
|
9 (50)
|
|
|
≤5 µg/dL
|
30 (46.2)
|
21 (44.7)
|
9 (50)
|
|
|
Tumor pathology
|
0.198
|
|||
|
ACTH + adenoma
|
55 (85)
|
40 (85.1)
|
15 (83.3)
|
|
|
Crooke degeneration
|
2 (3)
|
1 (2.1)
|
1 (5.6)
|
|
|
Pituitary hyperplasia
|
2 (3)
|
1 (2.1)
|
1 (5.6)
|
|
|
Normal pituitary tissue
|
6 (9)
|
5 (10.6)
|
1 (5.6)
|
|
|
Result of reoperation
|
0.740
|
|||
|
Remission
|
51 (78.5)
|
36 (76.6)
|
15 (83.3)
|
|
|
Residual disease
|
14 (21.5)
|
11 (23.4)
|
3 (16.7)
|
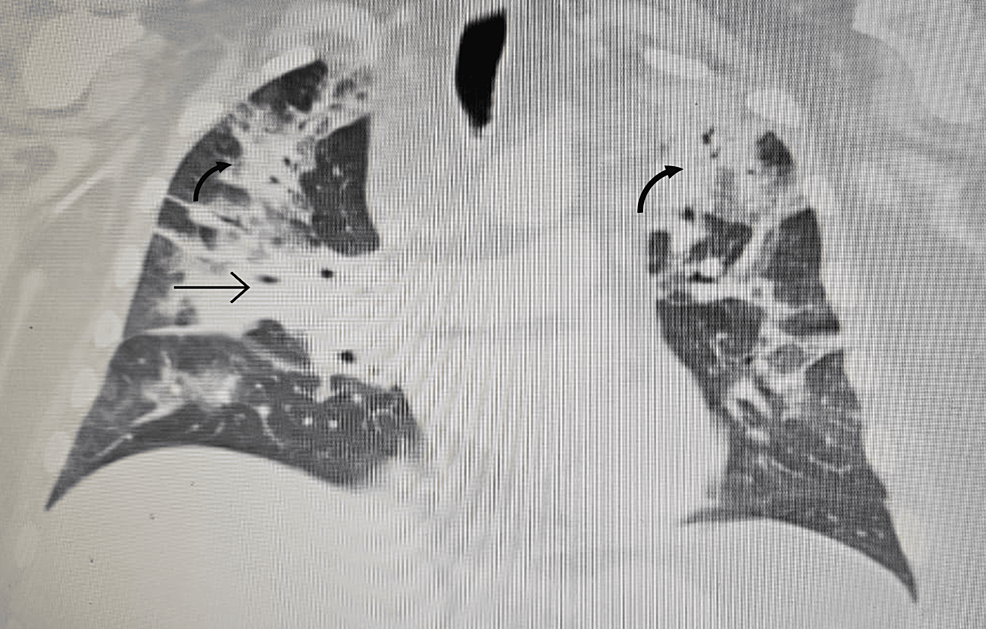
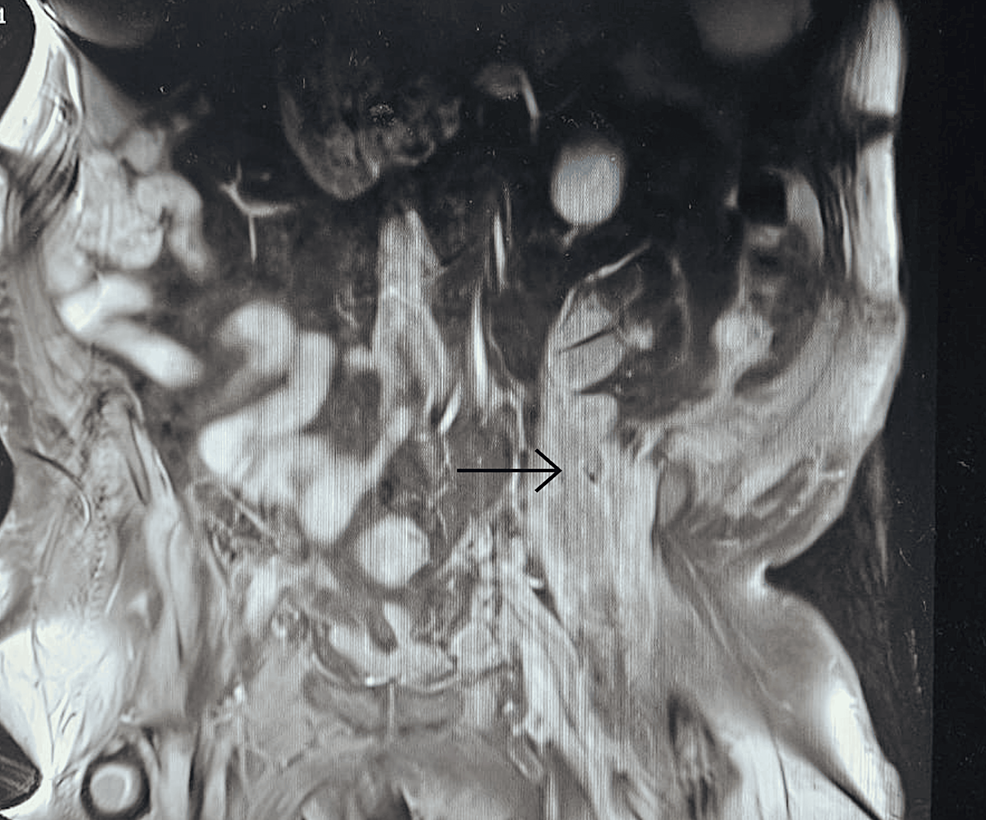
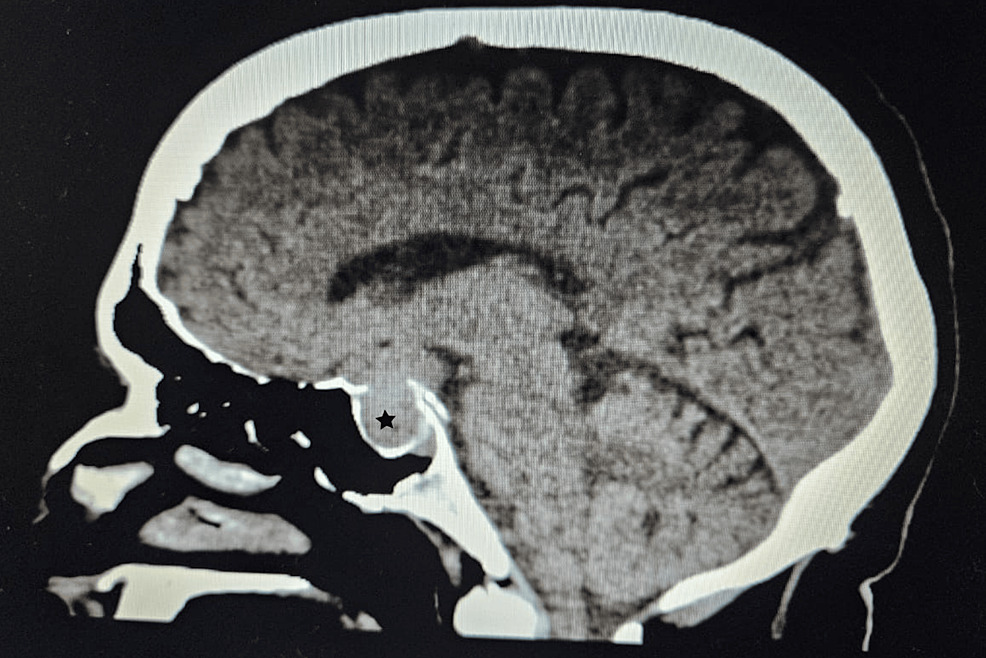
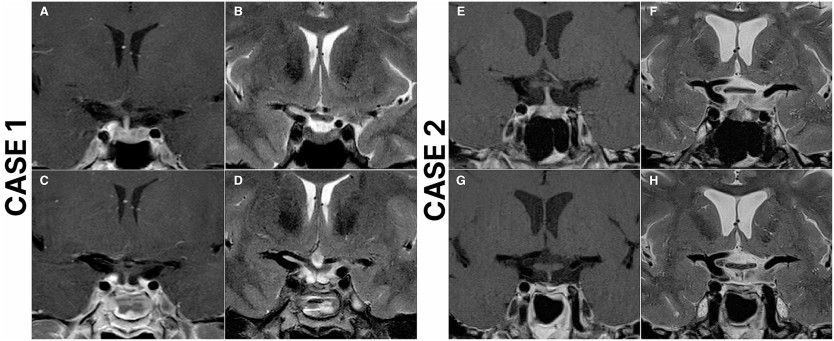
Radiological findings
Surgical characteristics
Outcomes
Factors predisposing to unsuccessful repeat endoscopic transsphenoidal surgery
Potential predictors of sustained remission
|
variable
|
OR (95% CI)
|
p -value
|
|---|---|---|
|
Age
|
1.003 (0.94–1.06)
|
0.913
|
|
Gender
|
||
|
Female
|
Reference
|
|
|
times
|
0.43 (0.06–2.88)
|
0.387
|
|
Indication for repeat ETS
|
||
|
Residual disease
|
Reference
|
|
|
Recurrence
|
1.2 (0.25–5.68)
|
0.812
|
|
Tumor size
|
||
|
Microadenoma
|
Reference
|
|
|
Macroadenoma
|
0.94 (0.18–4.79)
|
0.948
|
|
Relation to cavernous sinus
|
||
|
No relation
|
Reference
|
|
|
Extension invasion
|
0 (0)
|
0.999
|
|
Hardy-Wilson Classification
|
||
|
Degrees
|
||
|
I-II
|
Reference
|
|
|
III-IV
|
3.2 (0.3-34.06)
|
0.334
|
|
stage
|
||
|
AC
|
Reference
|
|
|
EN
|
0 (0)
|
0.999
|
|
POD 1 Cortisol levels
|
||
|
≤5 µg/dL
|
Reference
|
|
|
>5 µg/dL
|
0.09 (0.01–0.52)
|
0.006
|
Discussion
Strengths and limitations
Conclusion
Acknowledgments
Declarations
Ethical approval
Competing interests
Publisher’s Note
Filed under: Cushing's, pituitary, Treatments | Tagged: endonasal, endoscopic, neurosurgeon, recurrence, transsphenoidal | Leave a comment »