Investigations
Blood chemistry showed neutrophilic leucocytosis, hyperglycaemia with increased glycated haemoglobin, severe hypokalaemia and metabolic alkalosis (Table 1). Potassium infusion (50 mEq in 500 mL saline/24 h) was rapidly started, together with a subcutaneous rapid-acting insulin analogue and prophylactic enoxaparin. The patient experienced mental confusion, hallucinations and restlessness; non-enhanced computed tomography (CT) of the brain revealed a hypodense area of the splenium of the corpus callosum, possibly due to metabolic damage (Fig. 1A).
Table 1Hormonal and biochemical evaluation of patient throughout hospitalization and follow-up.
|
Normal range |
On hospital admission |
After surgery |
| 10 days |
2 months |
3 months |
6 months |
9 months |
12 months |
16 months |
| ACTH (pg/mL) |
9–52 |
551 |
7 |
37 |
50 |
29.5 |
26 |
40.9 |
52 |
| Morning cortisol† (µg/dL) |
7–19.2 |
63.4 |
14 |
5.1 |
3.5 |
3.8 |
4.2 |
7.2 |
12.8 |
| After 1 mg overnight dexamethasone |
|
|
|
|
|
|
|
|
|
| ACTH |
|
583 |
|
|
|
|
|
|
|
| Cortisol |
|
60 |
|
|
|
|
|
|
|
| DHEAS (µg/dL) |
9.4–246 |
201 |
|
|
|
|
|
|
|
| 24-h urinalysis (µg/24 h) |
|
|
|
|
|
|
|
|
|
| Adrenaline |
0–14.9 |
95.5 |
|
|
|
|
|
|
|
| Noradrenaline |
0–66 |
1133 |
|
|
|
|
|
|
|
| Metanephrine |
74–297 |
1927 |
|
|
|
|
|
|
|
| Normetanephrine |
105–354 |
1133 |
|
|
|
|
|
|
|
| Chromogranin A |
0–108 |
290 |
|
|
|
|
|
|
|
| Renin (supine) (µU/mL) |
2.4–29 |
3.9 |
|
|
|
14.6 |
|
|
|
| Aldosterone (supine) (ng/dL) |
3–15 |
3.4 |
|
|
|
12.5 |
|
|
|
| LH (mIU/mL)* |
> 10 |
0.3 |
|
|
|
65.8 |
|
|
|
| FSH (mIU/mL)* |
> 25 |
1.9 |
|
|
|
116 |
|
|
|
| PRL (ng/mL) |
3–24 |
13.7 |
|
|
|
|
|
|
|
| FT4 (ng/dL) |
0.9–1.7 |
1.1 |
|
|
|
1.2 |
|
|
|
| FT3 (pg/mL) |
1.8–4.6 |
1.1 |
|
|
|
2.7 |
|
|
|
| TSH (µU/mL) |
0.27–4.2 |
0.23 |
|
|
|
1.3 |
|
|
|
| PTH (pg/mL) |
15–65 |
166 |
|
|
|
|
|
|
|
| Calcium (mg/dL) |
8.2–10.2 |
8.2 |
|
|
|
|
|
|
|
| Calcitonin (pg/mL) |
0–10 |
1 |
|
|
|
|
|
|
|
| Glycaemia (mg/dL) |
60–110 |
212 |
69 |
|
73 |
|
|
|
83 |
| Potassium (mEq/L) |
3.5–5 |
2.4 |
3.3 |
3.9 |
4.2 |
3.7 |
5 |
4.4 |
3.9 |
| Leucocytes (K/µL) |
4.0–9.3 |
15.13 |
|
|
|
|
|
|
|
| HbA1c (mmol/mol) |
20–42 |
55 |
|
|
30 |
|
|
|
|
| HCO3− (mEq/L) |
22–26 |
41.8 |
|
|
|
|
|
|
|
The patient was transferred to the internal medicine ward. Although potassium infusion was increased to 120 mEq/day, serum levels did not normalize; a mineralocorticoid receptor antagonist (potassium canreonate) was therefore introduced, but the effect was partial. In order to control BP, the irbersartan dose was increased (300 mg/day) and amlodipine (10 mg/day) was added.
The combination of severe hypertension, newly occurring diabetes and resistant hypokalaemia prompted us to hypothesize a common endocrine aetiology.
A thorough hormonal array showed very high ACTH and cortisol levels, whereas supine renin and aldosterone levels were in the low-normal range (Table 1). Since our patient proved repeatedly non-compliant with 24-h urine collection, UFC could not be measured.
After an overnight 1 mg dexamethasone suppression test, cortisol levels remained unchanged, whereas ACTH levels slightly increased (Table 1). Notably, the patient showed no Cushingoid features. Gonadotropin levels were inappropriately low for the patient’s age; FT4 levels were normal, whereas FT3 and thyroid-stimulating hormone (TSH) levels were reduced and calcitonin levels were normal (Table 1). HbA1c levels were increased (Table 1).
Finally, secondary hyperparathyroidism, associated with low-normal calcium levels and reduced vitamin D levels, was found (Table 1).
Brain contrast-enhanced magnetic resonance (MR) imaging revealed a 5-mm median posterior pituitary microadenoma (Fig. 1B) and a hyperintense lesion of the splenium of the corpus callosum (Fig. 1C). Diffusion-weighted MR images of the lesion showed no restricted diffusion (Fig. 1D), thus excluding an ischaemic origin. Petrosal venous sampling for ACTH determination at baseline and after CRH stimulation was excluded, as it was deemed a high-risk procedure, given the patient’s poor condition.
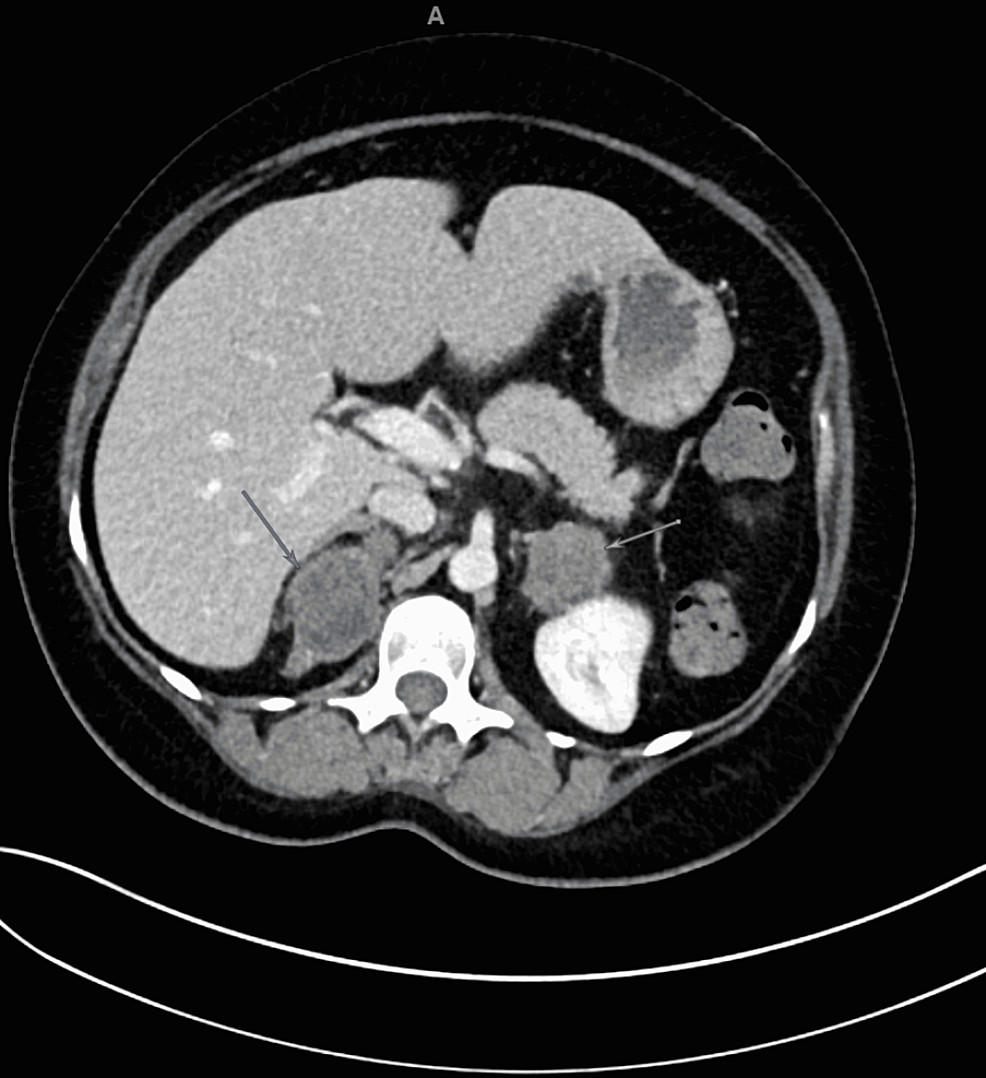
Since the ACTH and cortisol levels were greatly increased and were associated with severe hypokalaemia, EAS was hypothesized; total-body contrast-enhanced CT revealed the left adrenal mass (3 cm), which showed regular margins and heterogeneous enhancement (Fig. 2A and B) and measured 25 Hounsfield units. There was no evidence of adrenal hyperplasia in the contralateral adrenal gland. The adrenal mass showed intense tracer uptake on both 18F-FDG PET/CT (Fig. 2C and D), suggestive of adrenal malignancy or functioning tumour, and 68Ga-DOTATOC PET/CT (Fig. 3), which is characteristic of a neuroendocrine lesion. No other sites of suspicious tracer uptake were detected.
Bisoprolol was withdrawn, and 24-h urinary catecholamine, metanephrine and normetanephrine levels proved significantly increased, as were chromogranin A levels (Table 1). In sum, an ACTH-secreting pheochromocytoma was suspected and the pituitary microadenoma was deemed a likely incidental finding.
The patient’s mental state worsened, fluctuating from sopor to restlessness, which required parenteral neuroleptics and restraint. An electroencephalogram revealed a specific slowdown of cerebral electrical activity. Following rachicentesis, the cerebrospinal fluid showed pleocytosis (lympho-monocytosis), whereas a culture test and polymerase chain reaction for common neurotropic agents were negative. The neurologist hypothesized a psycho-organic syndrome secondary to severe metabolic derangement. Intravenous ampicillin, acyclovir and B vitamins were empirically started. The patient was transferred to the subintensive unit, where a nasogastric tube and central venous catheter were inserted, and enteral nutrition was started.
Discussion
The diagnosis of EAS is challenging and requires two steps: confirmation of increased ACTH and cortisol levels and anatomic distinction from pituitary sources of ACTH overproduction. Besides metabolic derangements (hyperglycaemia, hypertension), EAS-related severe hypercortisolism may cause profound hypokalaemia (3, 4, 5).
In our patient, the combination of worsening hypertension, newly occurring diabetes and resistant hypokalaemia raised the suspicion of a common endocrine cause.
ACTH-dependent severe hypercortisolism was ascertained, and subsequent brain MR revealed a pituitary microadenoma.
The diagnosis of CS requires the combination of two abnormal test results: 24-h UFC, midnight salivary cortisol and/or abnormal 1 mg dexamethasone suppression testing (2, 6). ACTH evaluation (low/normal-high) is fundamental to tailoring the imaging technique.
The very high cortisol levels found in our patient were unchanged after overnight dexamethasone testing, whereas UFC could not be assessed owing to the lack of compliance with urine collection. The accuracy of the UFC assays, however, may be impaired by cortisol precursors and metabolites. Salivary cortisol assessment was not performed since the specific assay is not available in our hospital.
The combination of ACTH-dependent severe hypercortisolism and hypokalaemia prompted us to suspect EAS. The differential diagnosis between pituitary and ectopic ACTH-dependent CS involves high-dose (8 mg) dexamethasone suppression testing, which has relatively low diagnostic accuracy (6). Owing to the patient’s very high cortisol levels and severe hypokalaemia, this testing was not performed, on account of the risks of administering corticosteroids in a patient already exposed to excessive levels (6). Furthermore, owing to the increase in ACTH levels observed after overnight dexamethasone testing, we postulated the possible occurrence of glucocorticoid-driven positive feedback on ACTH secretion, which has been described in EAS, including cases of pheochromocytoma (7).
Finally, in the case of EAS suspected of being caused by pheochromocytoma, we do not recommend performing high-dose dexamethasone suppression testing, owing to the risk of triggering a catecholaminergic crisis (8).
The dynamic tests commonly used to distinguish patients with EAS from those with Cushing’s disease are the CRH stimulation test and the desmopressin stimulation test, either alone or in combination with CRH testing (6). Owing to the rapid worsening of our patient’s condition, dynamic testing was not done; however, the clinical picture and hormonal/biochemical data were suggestive of EAS.
EAS is mainly (45–50%) due to neuroendocrine tumours, mostly of the lung (small-cell lung cancer and bronchial tumours), thymus or gastrointestinal tract; however, up to 20% of ACTH-secreting tumours remain occult (3, 4, 5).
ACTH-secreting pheochromocytomas are responsible for about 5% of cases of EAS (3, 4, 9, 10). Indeed, this rate ranges widely, from 2.5% (11) to 15% (12), according to the different case series. Patients with EAS due to pheochromocytoma present with severe CS, overt diabetes mellitus, hypertension and hypokalaemia (3); symptoms of catecholamine excess may be unapparent (3), making the diagnosis more challenging.
A recent review of 99 patients with ACTH- and/or CRH-secreting pheochromocytomas found that the vast majority displayed a Cushingoid phenotype (10); by contrast, another review of 24 patients reported that typical Cushingoid features were observed in only 30% of patients, whereas weight loss was a prevalent clinical finding (13). We hypothesized that the significant weight loss reported by our patient was largely due to the hypermetabolic state induced by catecholamines, which directly reduce visceral and subcutaneous fat, as recently reported (14).
Our patient showed no classic stigmata of CS, owing to the rapid onset of severe hypercortisolism (10, 13), whereas she had worsening hypertension and newly occurring diabetes mellitus, which were related to both cortisol and catecholamine hypersecretion; hypokalaemia was deemed to be secondary to severe hypercortisolism. Indeed, greatly increased cortisol levels act on the mineralocorticoid receptors of the distal tubule after saturating 11β-hydroxysteroid dehydrogenase type 2, leading to hypokalaemia (4). Consequently, hypokalaemia is much more common (74–95% of patients) in EAS than in classic Cushing’s disease (10%) (3, 4, 10). This apparent mineralocorticoid excess suppresses renin and aldosterone secretion, as was ascertained in our patient.
In this setting, the most effective way to manage hypokalaemia is to treat the hypercortisolism itself by administering immediate-acting steroidogenesis inhibitors, combined with potassium infusion and a mineralocorticoid receptor-antagonist (e.g. spironolactone) at an appropriate dosage (100–300 mg/day) (4).
In ACTH-secreting pheochromocytoma, cortisol hypersecretion potentiates catecholamine-induced hypertension by stimulating the phenol-etholamine-N-methyl–transferase enzyme, which transforms noradrenaline to adrenaline (4). Indeed, in our patient, the significant ketoconazole-induced reduction in cortisol secretion led to satisfactory BP control on antihypertensive drugs. After the biochemical diagnosis of pheochromocytoma, a selective alpha-blocker was added, and after a few days, a beta-blocker was restarted in order to control reflex tachycardia (15).
Our patient had greatly increased ACTH levels (>500 pg/mL) associated with very high cortisol levels (>60 µg/dL), which, together with the finding of hypokalaemia, prompted us to hypothesize EAS. With regard to these findings, ACTH levels are usually higher (>200 pg/mL) in patients with EAS than in those with CS due to a pituitary adenoma; however, considerable overlapping occurs (3, 11, 16). Most patients with ACTH-secreting pheochromocytomas in those series had ACTH levels >300 pg/mL, and a few had normal ACTH levels (9), thus complicating the diagnosis. In addition, patients with EAS usually have higher cortisol levels than those with ACTH-secreting adenomas (3, 11).
In our patient, the left adrenal mass was deemed the culprit of EAS, and owing to very high urinary metanephrine levels, a pheochromocytoma was suspected.
It can be assumed that the adrenal tumour, which was anamnestically reported as ‘non-secreting’, but on which only part of the initial hormonal data were available, was actually a pheochromocytoma at the time of the first diagnosis but displayed a silent clinical and hormonal behaviour. The mass subsequently showed significant uptake on both 18F-FDG PET/CT and 68Ga-DOTATOC PET/CT (4, 5). It is claimed that 68Ga-DOTATOC PET/CT provides a high grade (90%) of sensitivity and specificity in the diagnosis of tumours that cause EAS (4, 5); nevertheless, a recent systematic review reported much lower sensitivity (64%), which increased to 76% in histologically confirmed cases (17).
In patients with EAS, immediate-acting steroidogenesis inhibitors are required in order to achieve prompt control of severe hypercortisolism (4). Ketoconazole is one of the drugs of choice since it inhibits adrenal steroidogenesis at several steps. In our patient, ketoconazole rapidly reduced cortisol levels to normal values, without causing hepatic toxicity (4). Moreover, ketoconazole proved effective at a moderate dosage (600 mg/day), which falls within the mean literature range (18, 19). However, dosages up to 1200–1600 mg/day are sometimes required in severe cases (usually EAS) (18, 19). Speculatively, our results might reflect an enhanced inhibitory action of ketoconazole at the adrenal level, which was able to override the strong ectopic ACTH stimulation.
In addition, the finding that, following cortisol reduction, ACTH levels paradoxically decreased suggests an additive and direct effect of the drug. This effect has been observed in a few patients with EAS (20) and is supported by in vitro studies showing a direct anti-proliferative and pro-apoptotic effect of ketoconazole on ectopic ACTH secretion by tumours (21). Finally, the reduction in ACTH levels during treatment with steroidogenesis inhibitors prompts us to postulate the presence of glucocorticoid-driven positive feedback on ACTH secretion, as already described in neuroendocrine tumours (7, 20, 21). The coexistence of EAS and ACTH-producing pituitary adenoma is very rare but must be taken into account. In our case, we deemed the pituitary mass found on MR to be a non-secreting microadenoma. This hypothesis was strengthened by the finding that, following exeresis of the ACTH-secreting pheochromocytoma, ACTH normalized, hypercortisolism vanished and pituitary function recovered. These findings suggest that: (i) altered pituitary function at the baseline was secondary to the inhibitory effect of hypercortisolism; (ii) the excessive production of cortisol was driven by ACTH overproduction outside the pituitary gland, specifically within the adrenal gland tumour.
In our patient, a few days after surgery, morning cortisol levels before hydrocortisone bolus administration were ‘normal’. Owing to both the half-life of hydrocortisone (8–12 h) and the supraphysiological dosage used, it is likely that a residual part of the drug, which cross-reacts in the cortisol assay, was still circulating at the time of blood collection, thus resulting in ‘normal’ cortisol values. Following the switch to oral cortisone, cortisol levels before therapy were low, thus confirming post-surgical hypocortisolism. Hypocortisolism remained throughout the first year after surgery, and glucocorticoid therapy was continued. Sixteen months after surgery, baseline cortisol levels returned to the normal range; cortisone therapy was therefore tapered and a further hormonal check was scheduled. Assessment of the cortisol response to ACTH stimulation testing would be helpful in order to check the resumption of the residual adrenal function.
A peculiar aspect of our case was the occurrence of a psycho-organic syndrome together with the finding of a splenial lesion on brain imaging, which was deemed secondary to metabolic injury. Indeed, the increased cortisol levels present in patients with Cushing’s disease are detrimental to the white matter of the brain, including the corpus collosum, causing subsequent clinical derangements (22).
Besides the direct effects of hypercortisolism, the splenial damage was also probably due to long-standing hypertension, worsened by newly occurring catecholamine hypersecretion and diabetes. Together with the normalization of cortisol and glycaemic levels, and of BP, a partial reduction in the splenial damage was observed on two subsequent MR examinations, and the patient’s neurological condition slowly improved until she fully recovered.
In our patient, thorough germinal genetic testing for the commonest pheochromocytoma/paraganglioma (PPGL) genes proved negative. Since approximately 40% of these tumours have germline mutations, genetic testing is recommended regardless of the patient’s age and family history. In the absence of syndromic, familial or metastatic presentation, the selection of genes for testing may be guided by the tumour location and biochemical phenotype.
Alterations of the PPGL genes can be divided into two groups: 10 genes (RET, VHL, NF1, SDHD, SDHAF2, SDHC, SDHB, SDHA, TMEM127 and MAX) that have well-defined genotype–phenotype correlations, thus allowing to tailor imaging procedures and medical management, and a group of other emerging genes, which lack established genotype–phenotype associations; for patients in whom mutations of genes belonging to this second group are detected, and hence hereditary predisposition is established, only general medical surveillance and family screening can be planned (23, 24).
In conclusion, our case highlights the importance of investigating patients with hypertension and metabolic derangements such as diabetes and hypokalaemia, since these findings may be a sign of newly occurring EAS, which, in rare cases, may be due to an ACTH-secreting pheochromocytoma. Since the additive effect of cortisol and catecholamine can cause dramatic clinical consequences, the possibility of an ACTH-secreting pheochromocytoma should be taken into account in the presence of an adrenal mass. EAS must be considered an endocrine emergency requiring urgent multi-specialist treatment. Surgery, whenever possible, is usually curative, and anatomic brain damage, as ascertained in our patient, may be at least partially reversible.



 View Full Size
View Full Size View Full Size
View Full Size View Full Size
View Full Size View Full Size
View Full Size View Full Size
View Full Size