A Puerto Rican woman aged 50 years presented to an ophthalmologist with complaints of vision changes, including difficulty seeing images in her peripheral vision in both eyes and difficulty in color perception. Her medical history was significant for menopause at age 43 years, type 2 diabetes and hypertension. She had no prior history of thyroid disease, changes in her weight, dizziness or lightheadedness, headaches, galactorrhea or growth of her hands or feet.
Formal visual fields showed bitemporal superior quadrantopsia, and she was sent to the ED for further evaluation.
Imaging and laboratory tests
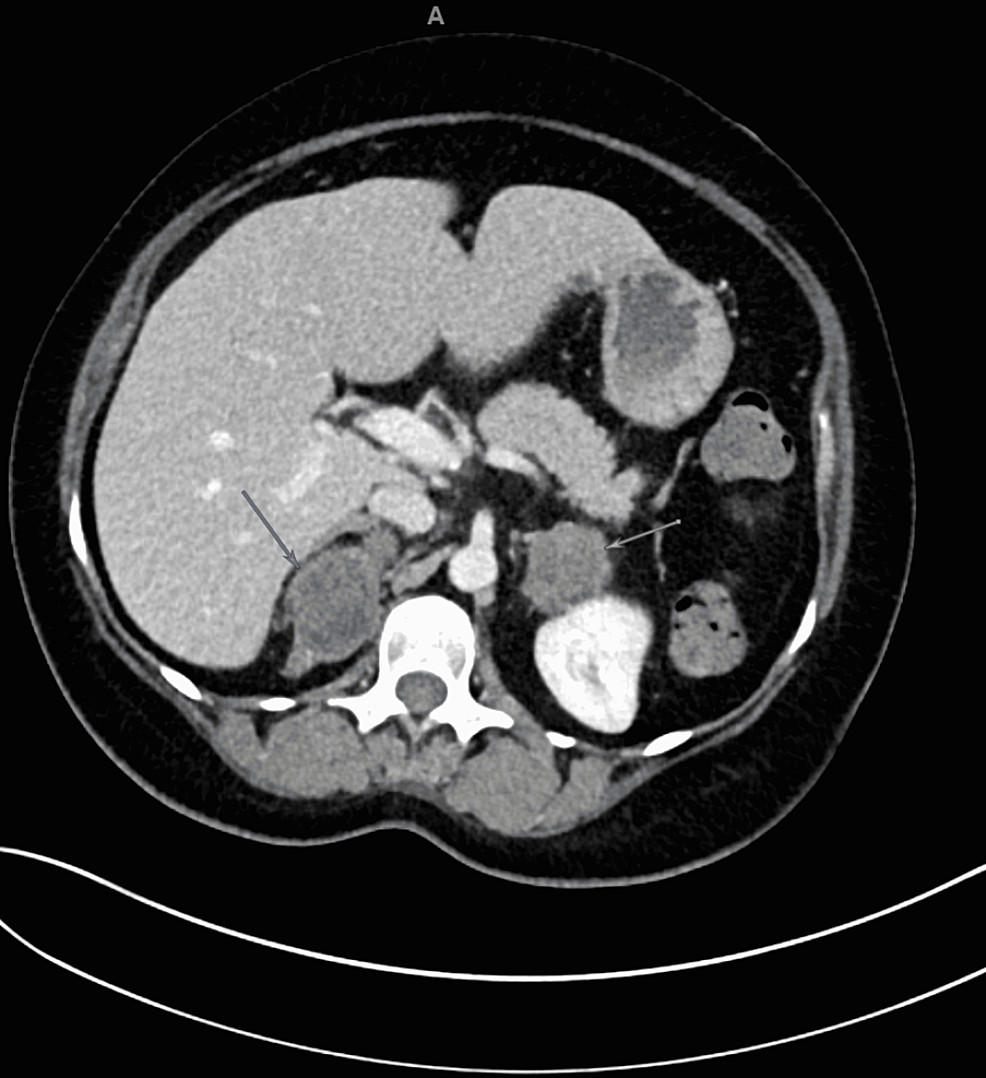
A pituitary protocol MRI was performed that showed a large 3 cm x 2 cm x 2.2 cm mass in the pituitary with mild osseous remodeling of the sella turcica and mass effect on the optic chiasm (Figure 1). The mass was isointense with the brain parenchyma on T1-weighted and T2-weighted images and homogeneously enhanced after IV gadolinium contrast administration.
Baseline laboratory samples drawn at 11 p.m. in the ED showed a cortisol of 16.9 µg/dL (nighttime reference range: 3-16 µg/dL), adrenocorticotropic hormone 65 pg/mL (reference range: 6-50 pg/mL), prolactin 19.4 ng/mL (reference range: 5.2-26.5 ng/mL), thyroid-stimulating hormone 1.36 µIU/mL (reference range: 0.35-4.9 µIU/mL), free thyroxine 0.9 ng/dL (reference range: 0.6-1.8 ng/dL), triiodothyronine 85 ng/dL (reference range: 83-160 ng/dL), follicle-stimulating hormone (FSH) 11.1 mIU/mL (postmenopausal reference range: 26.7- 133.4 mIU/mL) and luteinizing hormone (LH) 1.2 mIU/mL (postmenopausal reference range: 5.2-62 mIU/mL).
Figure 1. T1-weighted MRI images with and without contrast of the pituitary. Coronal (A) and sagittal (C) images showed a large isodense (with brain parenchyma) 3 cm x 2 cm x 2.2 cm mass (red arrow) in the sella with superior extension to the optic chiasm. After gadolinium contrast, coronal (B) and sagittal (D) images show the mass homogenously enhances consistent with a pituitary adenoma.
Images courtesy of Pavani Srimatkandada, MD.
Given the patient’s high nighttime cortisol and adrenocorticotropic hormone (ACTH) levels, she underwent an overnight dexamethasone suppression test with 1 mg dexamethasone. Her morning cortisol was appropriately suppressed to less than 1 µg/dL, excluding Cushing’s disease.
Pituitary adenoma resection
The patient was diagnosed with a nonsecreting pituitary adenoma with suprasellar extension and optic chiasm compression with visual field deficits. The macroadenoma caused an inappropriately normal LH and FSH in a postmenopausal woman consistent with hypogonadotrophic hypogonadism.
She underwent transnasal transsphenoidal resection of the nonsecreting pituitary adenoma. The dural defect caused by the surgery was patched with an abdominal fat graft with a DuraSeal dura patch. A postoperative MRI showed complete resection of the adenoma with no evaluable tumor in the sella (Figure 2). Her postoperative course was complicated by transient diabetes insipidus requiring intermittent desmopressin; however, this resolved before her discharge from the hospital.
Figure 2. T1-weighted MRI images with contrast. Coronal views before (A) and after (B) transphenoidal tumor resection show complete resolution of the enhancing pituitary mass (A; red arrow) that is replaced with a new hypodense mass in the sella (B; yellow arrow). This mass is filled with cerebrospinal fluid with a residual rim of enhancing tissue. This is consistent with the development of a pseudomeningocele in the sella.
Postoperative testing confirmed secondary deficiency of the adrenal, thyroid and ovarian axes requiring hormone therapy. The patient had stable temporal hemianopia in the left eye with improved vision in the right eye.
Recurrent mass detected
One year after surgery, during a routine follow-up appointment, the patient reported no dizziness, lightheadedness, worsening vision changes, rhinorrhea or headache. She had a follow-up MRI of the brain with and without contrast, which showed the interval appearance of a mass in the sella that extended from the sphenoid sinus into the sella and came in contact with the optic nerve (Figure 3).
Figure 3. Axial MRI images of the sella after resection of pituitary adenoma. On T1-weighted images the mass (red arrow) in the sella is hypodense (black) compared with the brain parenchyma. On T2-weighted images, the mass (red arrow) is hyperdense (bright) compared with the brain, consistent with fluid. Cerebrospinal fluid in the sulci on the brain surface and the vitreous fluid within the eye are also hyperintense on T2-weighted images (yellow arrows).
On MRI, the mass was isodense with the cerebral spinal fluid (CSF) with a residual rim of enhancing normal pituitary tissue. This appearance is consistent with the postoperative development of a pseudomeningocele and not a solid mass in the sella (Table).
Pseudomeningoceles are abnormal collections of CSF that communicate with the CSF space around the brain; these occur after brain surgery involving duraplasty (incision and repair of the dura). Unlike meningoceles, pseudomeningoceles are not completely encased by a surrounding membrane, and they communicate with the circulating CSF. Similar to CSF, a pseudomeningocele is hypodense (dark) compared with brain on T1-weighted MRI images and hyperdense (bright) on T2-weight images.
Pseudomeningocele treatment
Treatment may be conservative or may involve neurosurgical repair if symptomatic. Little published data addresses the development of pseudomeningoceles after transsphenoidal pituitary surgery, but this complication occasionally occurs, especially if the dural incision is large. One study noted that pseudomeningoceles are one of the most common complications after suboccipital decompression for Chiari’s malformation, but the effect of this complication is unclear.
Endocrinologists must recognize that recurrent development of pituitary masses after transsphenoidal pituitary adenoma surgery may not represent regrowth of pituitary tissue, but instead development of a meningocele/pseudomeningocele. Pseudomeningocele can be easily confirmed because this fluid collection has very different MRI characteristics than pituitary adenoma (Table). Given that patients may remain asymptomatic after the development of a pseudomeningocele, periodic MRI imaging, hormonal evaluation and ophthalmologic monitoring of visual fields are required after transsphenoidal pituitary surgery.
References:
- Hernandez Guilabert PM. Poster No C-1330. Presented at: European Society of Radiology; March 7-11, 2013; Vienna.
- Parker SL, et al. J Neurosurg. 2013;doi:10.3171/2013.8.JNS122106.
For more information:
- Stephanie L. Lee, MD, PhD, ECNU, is an associate professor of medicine and associate chief, in the Section of Endocrinology, Diabetes and Nutrition at Boston Medical Center. Lee can be reached at Boston Medical Center, 88 E. Newton St., Endocrinology Evans 201, Boston, MA 02118; email: stephanie.lee@bmc.org. Lee reports no relevant financial disclosures.
- Pavani Srimatkandada, MD, is an endocrinology fellow in the Section of Endocrinology, Diabetes and Nutrition at Boston Medical Center. Srimatkandada can be reached at Boston Medical Center, 88 E. Newton St., Endocrinology Evans 201, Boston, MA 02118. She reports no relevant financial disclosures.
From http://www.healio.com/endocrinology/thyroid/news/print/endocrine-today/%7B82430fb6-bbe4-4908-a389-447eee8cd005%7D/recurrent-sellar-mass-after-resection-of-pituitary-macroadenoma
Filed under: pituitary, Rare Diseases, Treatments | Tagged: ACTH, Adrenocorticotropic hormone, cerebral spinal fluid, cortisol, CSF, dexamethasone suppression test, dizziness, galactorrhea, headaches, lightheadedness, menopause, pituitary, sella turcica, thyroid, transsphenoidal, Type 2 diabetes, vision, weight | Leave a comment »








 Radiation therapy can be effective in controlling the growth of the tumor. However, if you received radiation therapy in the past, additional radiation may not be safe.
Radiation therapy can be effective in controlling the growth of the tumor. However, if you received radiation therapy in the past, additional radiation may not be safe. Medical therapies for the treatment of Nelson’s syndrome are currently limited, but include:
Medical therapies for the treatment of Nelson’s syndrome are currently limited, but include:


